| |
HER2 Testing and Trastuzumab |
EDITOR’S COMMENT
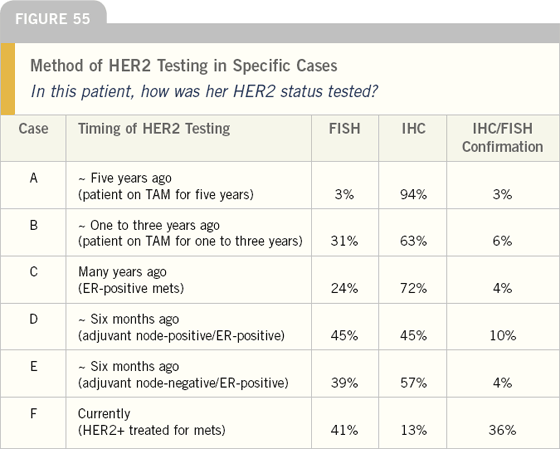
One of the fascinating and unexpected findings of this survey is related to HER2 testing. We asked these physicians to select six patients from their practices in a variety of situations. As a routine part of the data collection, we asked whether the technique utilized was immunohistochemistry (IHC) or fluorescence in situ hybridization (FISH). When we first looked at the results, we saw a surprising heterogeneity that was difficult to explain. However, upon further study, what we saw seemed to correlate with both the likely timing of the testing and the rationale for it. Almost all women who were originally diagnosed more than one year ago had undergone IHC testing, but FISH was more commonly used in women diagnosed in the last year (and having adjuvant decisions made). For patients in whom the use of trastuzumab was being considered, FISH testing was utilized exclusively.
Related Comments from Research Leaders
I assume that the tumors with a 3+ score on IHC are truly HER2- positive, and we do not test them further. An IHC score of 3+ is pretty reliable, as long as it is done at a laboratory that performs a lot of assays. If a tumor has a 2+ score on IHC, we test with FISH. Even in patients with an IHC score of 0 or 1+ and other features of excessively aggressive disease, we may do a FISH test.
Both the Intergroup and the NSABP study discovered that smaller community hospitals were overscoring tumors as 3+. Close to 20 percent of the 3+ scores were downstaged when they were reviewed centrally. The Intergroup protocol has now been amended to require that the patients wait for final randomization until there is a central review of their HER2 status.
I think the same things apply to FISH testing. Since FISH testing already tends to be done at more centralized laboratories, we have not yet explored the quality control issues. I suspect there will be a proliferation of FISH testing, and the reagents will go out to all the community hospitals. Even though there is probably less room for interobserver variability, the same issues will apply. I hope as the FISH technology disseminates, people will do these quality control-type studies.
— Debu Tripathy, MD
Whenever we have a new therapy requiring a predictive test, how that therapy performs is dependent on how good the test is at identifying the appropriate target. Both the NSABP adjuvant trial and the Intergroup trial indicated that HER2 testing in centers around the country — both community centers and academic centers — appeared to be less than perfect. Approximately 25 percent of the time, the test that was done in the local hospital — nonacademic institutions and academic institutions alike — couldn’t be confirmed at a central testing site.
We need to be careful about where the HER2 testing is performed and view results from less-experienced labs with caution. This is especially important in the adjuvant setting where, unlike the metastatic setting, we have no way of knowing if the treatment is working, and we’re committing the patient to a course of therapy.
Also, when we are banking on results from clinical trials, it is critical that we know the testing is accurate. Currently, trastuzumab has no established adjuvant role, but I suspect in the next three to five years we’ll learn whether it’s an effective adjuvant therapy. Then, accurate testing will be important to correctly identify the patients who will receive the maximum benefit from therapy.
In metastatic disease when the initial HER2 test results and the clinical situation are inconsistent, one should consider retesting the patient. I’ve had a number of patients whose tumors were IHC zero, but their clinical presentation was consistent with HER2 amplification, so I retested. In each one of those cases there was not a discrepancy, but still I think it’s worth doing. Even if I find a discrepancy only two out of 100 times, I’m doing those two patients a huge service.
— Eric P Winer, MD
Community laboratories don’t have the same performance when compared to the “gold standard” of commercial reference laboratories. Therefore, it is important to find out who is doing the HER2 testing. Good clinicians can also take other clinical variables into account to decide about retesting.
A good deal of evidence shows a correlation between the number of cases one analyzes per week with IHC and assay performance, and that’s where commercial laboratories win hands down. They do many more tests per week than a small hospital in rural North America. The bottom line is that there is a learning curve with respect to reading IHC stains. To get to the top of the curve you have to read a lot of them, and the only way to do that is to be in a in a big, busy center or in a commercial laboratory.
— Mark Pegram, MD
After laboratories underwent training from the NSABP and became certified, their accuracy went way up. Several things can be done to improve performance and reduce variability. One is to train the interpreter; another is to have the laboratory certified. It’s very important that laboratories participate voluntarily in these quality control programs and that they use controls with every assay.
Oncologists need to be more aware of which laboratory performs the tests and who interprets the results, because it can make a huge difference. Whether it’s a hospital-based laboratory or a reference laboratory, I think the oncologist should spend a lot of time getting to know their laboratories, which tests they’re using and how they read the results and interpret oncology and pathology guidelines.
— Ann D Thor, MD
The NSABP found the discordance rate to be much lower when experienced or certified laboratories are used for HER2 testing. This is really good for clinical care, because HER2 testing is not only being done for patients potentially eligible for clinical protocols, but also in general clinical practice.
— Edith Perez, MD
IHC was all we initially had available for testing, but early on we saw that IHC was flawed. IHC has a false-negative rate of about 18 percent. In a good laboratory, the false-positive rate for IHC is probably a few percent; it goes up to eight percent in general laboratories and was as high as 40 percent in some of the early reported trials.
Mike Press has data demonstrating a 52 percent concordance with the Dako HercepTest™ among Dako-approved pathologists. The College of American Pathologists has done its own study evaluating the concordance between a central laboratory and pathologists in the community. They are seeing similar trends.
— Dennis Slamon, MD, PhD
In our experience, it is highly unusual for the HER2 status to be altered during the development of the cancer. It is also very rare for us to find disagreement between the HER2 status of the invasive disease and the carcinoma in situ in the same patient. This is also true when we compare the primary tumor to the lymph-node metastasis.
In general, the HER2 status is quite similar or the same with only rare exceptions. In some of those exceptions, the morphologic appearance of the metastasis appears to be different, as if the tumor either developed new characteristics or was developed from an independent primary tumor.
— Michael F Press, MD, PhD
If one wants to know whether a patient has the HER2 alteration, one should do FISH testing. One should not do a default IHC and only if the tumor scores 2+, then do FISH. Using that algorithm, patients without the HER2 alteration will be treated with trastuzumab, and other patients with the HER2 alteration may not be treated.
The BCIRG trial we are conducting was designed with FISH as the only criteria for assessing HER2 status. I think the day is coming when FISH testing is the only assay used in the community, and I hope it will be sooner rather than later.
— Dennis Slamon, MD, PhD
Every patient with metastatic breast cancer in my practice has her tumor evaluated for HER2 gene amplification by FISH. Tumors with an IHC score of 3+ should be evaluated by FISH, because they may not have gene amplification. In tumors with an IHC score of 0 or 1+, three percent and seven percent, respectively, will have HER2 gene amplification by FISH. We need to determine HER2 status accurately, because it is a matter of life or death.
— Melody A Cobleigh, MD
To determine a patient’s HER2 status, FISH is currently the best method we have in terms of linking outcome with intervention. I believe ascertaining the HER2 status in patients with metastatic breast cancer is mandatory. One can use the primary tissue; however, whenever feasible, one should biopsy metastatic lesions and re-evaluate the HER2 and hormone receptors.
– Nicholas J Robert, MD
EDITOR’S COMMENT
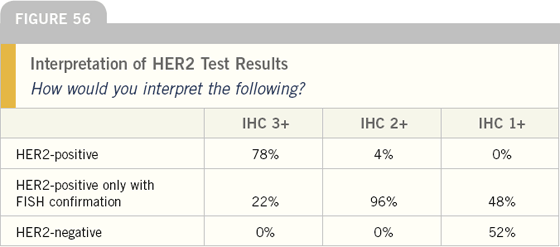
When queried about their algorithm for HER2 assessment, most physicians employed the approach commonly stated by research leaders — an IHC of 3+ is considered positive, and any other result requires FISH confirmation. However, 22 percent of oncologists require FISH testing even for tumors that are IHC positive.
Related Comments from Research Leaders
We were surprised when we found poor concordance between community and central laboratory testing, in terms of both HER2 protein expression and gene amplification. Perhaps more unexpected, we found poor concordance in terms of FISH testing in a central laboratory compared to the local laboratories. This last fact really came as a surprise, because the prevalent notion regarding FISH was that it was 100 percent accurate.
The data from these 119 cases were so important that we actually changed the eligibility criteria for this large cooperative group trial (NCCTG-N9831). We modified the protocol so that physicians can still conduct HER2 testing based on any technology in their local laboratories. The patient is then enrolled in the study and starts the doxorubicin/ cyclophosphamide (AC) portion of the chemotherapy.
During that time, we test the tumor specimens again by the HercepTest™ and the PathVysion™ FISH assay. If we find that neither of those two tests demonstrates HER2 positivity, we send the tumor specimen to another central laboratory to double-check our laboratory at the Mayo Clinic. If the other central laboratory also finds the tumor HER2-negative by both assays, then we notify the physician that the patient really should not participate in the trial.
— Edith Perez, MD
Considerable controversy remains regarding the optimal method to routinely evaluate HER2 status. I won’t treat a patient with metastatic breast cancer until I have a FISH assay.
In the June 2002 issue of the Journal of the National Cancer Institute, the NSABP and the Intergroup published their experiences with HER2 assessment, and it really cast doubt about our quality control for immunohistochemistry. Until the College of American Pathologists does something to iron out this problem of quality control, I continue to use FISH.
— Charles L Vogel, MD, FACP, PA
Tumors that score 2+ IHC are frequently found to be HER2-negative when tested by FISH. In those patients, I routinely have their tumors retested by FISH. On the other hand, I do not obtain a FISH analysis for tumors that score 3+ on IHC performed at a laboratory where I trust the pathologist.
Because HER2-positive breast cancer has a fairly specific phenotype (ie, steroid receptor-negative, younger age, early relapse), I will retest those types of patients by FISH if I have a two- to three-year-old IHC score of 0 or 1+. If the patient’s tumor is IHC-negative and FISH-positive, I treat them with trastuzumab despite the fact that we do not have clinical data for that group of patients. Tumors that are FISH-positive are likely to have ample amounts of HER2 receptors on their cell surface.
We lack quality control for both IHC and FISH. This is analogous to the situation encountered with estrogen receptor testing in the mid- to late 1970s. One wonders how many patients died because they did not receive adjuvant tamoxifen as a result of inadequate estrogen receptor testing. If adjuvant trastuzumab provides a benefit like adjuvant tamoxifen, we may encounter the same problem.
— George Sledge, MD
It really looks like IHC testing should remain in the purview of central reference laboratories. One of the reasons may be that a number of the large reference laboratories are now using digital image analysis for all of their IHC scoring. Digital image analysis takes some of the guesswork out of the interpretation of these IHC assays. Good pathologists can disagree over the difference between a 2+ and a 3+, but a computer can actually read the same slide over and over again and give you the exact same result.
Pathologists actually call up the information on a digital screen to confirm and double check the assay performance. In most of the large studies in which headto- head comparisons have been done with digital image analysis and FISH for HER2 testing, the concordance rate is about 90 percent.
— Mark Pegram, MD

EDITOR’S COMMENT
Trastuzumab monotherapy has been demonstrated to have significant antitumor activity and is frequently utilized in patients with asymptomatic metastases; however, most oncologists in this survey combine trastuzumab with chemotherapy, particularly in patients with symptoms. Surprisingly, a small yet substantial minority of oncologists do not utilize trastuzumab as part of first-line therapy for these patients even though essentially every research leader would support its incorporation.
Related Comments from Research Leaders
I tend to put patients into three categories— low risk, intermediate risk and high risk. I look at the low-risk category as an opportunity to give trastuzumab by itself. As the risk increases, I add more agents. My double-agent combination has generally been a taxane and trastuzumab, while my three-drug combination has been taxane/platinum/ trastuzumab.
If a patient is fairly asymptomatic and doesn’t have much disease, I offer her trastuzumab by itself and see how it goes. Anecdotally, I have had some patients do very well with trastuzumab monotherapy. We conducted a trial in which patients had the opportunity to have a lead-in induction with trastuzumab. Patients who had stable disease or better remained on trastuzumab for eight weeks and then received an additional eight weeks of treatment.
In patients who had evidence of progressive disease, paclitaxel and carboplatin were added to the trastuzumab. It was a small trial of 63 patients, but if you look back and see how the patients fared, we didn’t lose any ground during those first eight weeks in patients who didn’t benefit from trastuzumab.
For a patient who clearly has visceral metastases and is symptomatic, I use the three-drug combination with the platinum included. The other patients fall in the mix, and we discuss which one to start with and how aggressive to be.
— Howard A Burris III, MD
The decision to use trastuzumab sequentially versus concomitantly with chemotherapy is based on issues such as extent of metastatic disease and the time between diagnosis and progression. In a younger, relatively asymptomatic patient with bone metastases and a good performance status, I don’t think compelling evidence exists to use both chemotherapy and trastuzumab initially. No randomized trial compares sequential versus concomitant therapy in such a patient, but in other settings comparing sequential versus concomitant therapy with chemotherapy, concomitant therapy doesn’t do any better in terms of survival.
Certainly we enounter patients with metastatic disease in whom we feel chemotherapy is indicated, such as patients with significant visceral or life-threatening disease. Given the positive results of the trials in which trastuzumab was added to chemotherapy — improved response rate, time to progression and survival — my approach has been to give trastuzumab with the chemotherapy. Given our recent Phase III trial results, I would use the carboplatin/paclitaxel regimen.
— Nicholas J Robert, MD
If a woman has a hormone receptornegative tumor, the only strategy we have is chemotherapy, but if the tumor is HER2-positive, then I give chemotherapy with trastuzumab. Oncologists who prefer to begin with an anthracycline- based regimen as first-line therapy for a HER2-positive tumor presumably do so because of a historic belief that everyone needs an anthracycline up front. I don’t believe that’s true.
We now have a variety of active nonanthracycline-based drugs, and trastuzumab has clearly been shown to improve survival. I think that’s the priority, and we should rely on that data rather than falling back on data from the 1970s.
Several published trials showed the response rate to single-agent trastuzumab is on the order of 30 to 35 percent in patients whose tumors are HER2 3+ by IHC or FISH-positive, so monotherapy is a viable option. However, the response rates to chemotherapy plus trastuzumab are typically twice that, so I usually start with a combination.
— Harold J Burstein, MD, PhD
Next page
Page 1 of 2
|

