|
EDITOR’S COMMENT
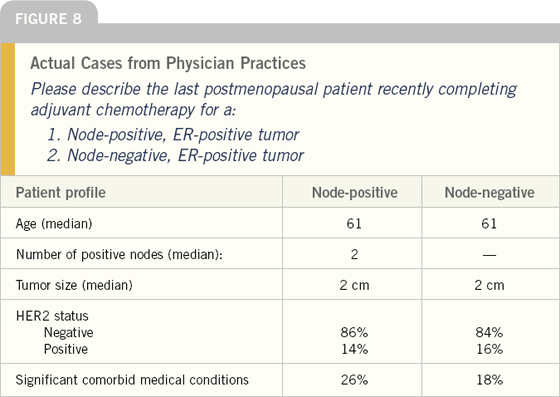
In many prior telephone surveys of physicians, we have posed theoretical case scenarios and asked oncologists how they might treat such patients. For this project, many similar questions were asked. However, we also added a new component in which physicians were queried about the last breast cancer patient they managed in very specific situations. The goal was to obtain “real world” insight into how patients are managed. The first two scenarios involved postmenopausal patients with ER-positive tumors who had recently completed adjuvant chemotherapy. We asked for one case of a patient with a node-positive tumor and another that was node-negative. The median age of 61 for these patients is lower than expected, perhaps reflecting a reluctance to use chemotherapy in older women. For patients with node-positive tumors, the median number of nodes was two, in keeping with the stage migration of breast cancer related to extensive use of mammographic screening. Note that a substantial number of treated patients had comorbid medical conditions that might have affected treatment decisions.
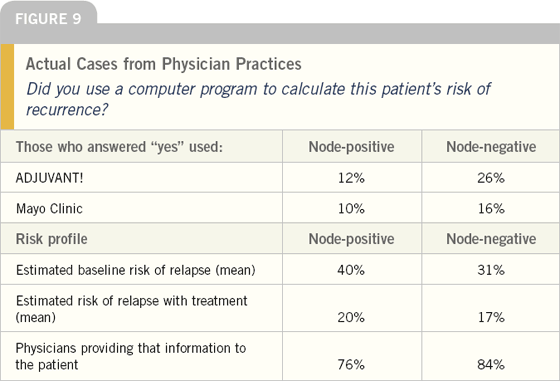
In recent years, two important computer web-based models have been developed to assist physicians in estimating the risk of recurrence and mortality in the adjuvant setting. Forty percent of physicians surveyed stated that they have used the Ravdin model and 21 percent have used the Mayo Clinic model. For these two cases, we found that the models were used more often in women with nodenegative tumors, probably because the results were important in determining whether chemotherapy would be utilized. More than three-fourths of physicians stated that they provided this information to these patients. Note also that the estimated risk for relapse was similar in node-positive and node-negative cases, reflecting that only women with nodenegative tumors at higher risk were likely to have been treated.
Related Comments from Research Leaders
The overview suggests that the proportional benefits hold up for a given therapy, irrespective of the baseline risk. If patients at low risk benefit 20 percent from a given therapy, patients at high risk receive a 20 percent relative benefit as well. So for the patient at low risk, a 20 percent benefit may be only one or two percent. But for the patient at high risk (50 percent), a 20 percent difference is a 10 percent risk reduction. This therapeutic index gets higher and higher with risk.
— Peter Ravdin, MD, PhD
The ADJUVANT! computer program (available at http://www.adjuvantonline.com) is based on nearly a decade of work that originated with information from the San Antonio database. The underpinnings of ADJUVANT! were based on the Surveillance, Epidemiology and End Results (SEER) database, a large population-based database with some great strengths. I believe it is a relevant database, and it’s certainly more population-based than the three percent of patients who participate in clinical trials. However, some approximations about the impact of adjuvant therapy in different groups must be made, because information about them is not included in SEER.
ADJUVANT! focuses on the baseline data for untreated patients, and it also incorporates the Overview’s data about the efficacy of different adjuvant therapies. The Overview approximates the absolute benefit by multiplying baseline estimates and proportional risk reductions.
One of the strengths of ADJUVANT! is its extensive help files which describe the assumptions that underlie some of the estimates. As much as possible, the assumptions in ADJUVANT! are based on global composite information, like the Overview.
— Peter Ravdin, MD, PhD
Oncologists, in conjunction with their patients, make decisions regarding adjuvant systemic therapy for primary breast cancer every day. Such decisions need to be individualized based on the characteristics of the primary tumor and the willingness of the patient to undergo toxicities for potential benefits.
When asked about how treatment decisions are made, oncology experts routinely reply that patients need to be informed of the options and that they need to participate in the decision-making process.
The question at hand is: How do physicians best inform themselves and their patients regarding the potential benefits associated with adjuvant systemic therapy for primary breast cancer? In a survey of women who had previously received adjuvant chemotherapy, only a minority of women remembered receiving any estimates regarding their prognosis with or without adjuvant systemic therapy, thus suggesting that there is room for improvement in providing patients with adequate information on adjuvant therapy.
— Loprinzi CL, Thome SD.
J Clin Oncol 2001;19(4):972-9.
Significant advances in the treatment of breast cancer have been made during the last several decades through the conduct of large, prospective, randomized clinical trials. These trials offer clinicians clear estimates of cancer mortality risk and survival benefit with the addition of chemotherapy. However, the wealth of clinical trial data has been accompanied by increasing complexity of decision-making and greater confusion among patients as a result of the myriad options available.
This is especially true for postmenopausal patients with estrogen receptorand/ or progesterone receptor-positive tumors for whom chemotherapy may add marginal extra benefit in addition to hormonal therapy with tamoxifen.
Obtaining accurate information regarding the risks and benefits of all treatment options is an integral part of the patients’ decision-making process and subsequent informed consent. In addition, physician influence has been shown to have a great effect on patient preferences for treatment options.
— Chao C et al.
J Clin Oncol 2003;21(23):4299-305.
Ideally, the patient should be given the chance to make the decision about whether to receive adjuvant chemotherapy, using the information oncologists provide. Although some patients will want adjuvant chemotherapy for even a 1% improvement in cure rate, others may find a 5% to 10% absolute improvement in cure rate not worth the short-term side effects of chemotherapy.
However, no matter what the relative benefit of adjuvant chemotherapy is, it behooves oncologists to participate in an honest discussion of the benefits and risks of chemotherapy in every individual patient and give the patient the chance to make the decision for, or against, adjuvant chemotherapy.
— Sonpavde G.
J Clin Oncol 2003;21(5):948-9.
An absolute 1% risk reduction may seem low to some readers, especially when the treatment given is chemotherapy. However, interviews of cancer patients show that a sizable proportion of them (up to one half of them in one study) consider a 1% chance of cure as a valid justification for undertaking chemotherapy.
Patients also would accept a hormone therapy for low expected benefits. Age does not seem to alter the willingness to accept treatment, although older patients may have a somewhat higher threshold in survival benefit to accept the most toxic treatment alternatives.
In the EBCTCG meta-analysis, the absolute decrease in mortality with chemotherapy for node-positive women aged 50 to 69 years was 2.3% at 10 years. Such a well-accepted intervention as beta-blockers after a myocardial infarction leads to an absolute 1.8% decrease in long-term mortality.
Similarly, prolonged antiplatelet therapy for various cardiovascular conditions yields a 2% to 5% long-term decrease in mortality.
Therefore, the 1% to 3% absolute reduction in mortality risk that elderly breast cancer patients can expect from chemotherapy according to this model is within the range of effectiveness of common secondary prevention interventions. Given the prevalence of breast cancer in older people, such a degree of benefit translates into a significant impact from a population perspective.
— Extermann M et al.
J Clin Oncol 2000;18(8)1709-17.
EDITOR’S COMMENT
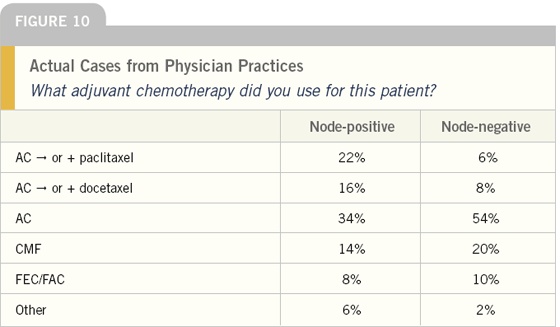
One of the most surprising findings in this survey was selection of chemotherapy in postmenopausal women with node-positive tumors. In other surveys — including this one — oncologists have claimed that they typically administer a taxane to these patients. However, in this scenario in which the physicians were queried about the last specific patient they evaluated, less than half of the patients received a taxane. This important clue from our pilot effort will now be rigorously evaluated in a more definitive survey with more physicians and greater statistical power.
If the findings hold up, a number of potential explanations could apply, including the perception that taxanes do not provide incremental benefit in women with receptorpositive tumors. Another possible explanation is that based on models like ADJUVANT!, the incremental absolute advantage to taxanes in women already receiving an aromatase inhibitor might not justify utilization. Another surprise was that only about a third of physicians using AC followed by T in these cases delivered therapy with growth factors every two weeks — the dose-dense approach that CALGB-9741 demonstrated to result in an improved survival compared to non-dose-dense AC followed by T.
Related Comments from Research Leaders
We now have a number of adjuvant regimens that are better than the standard regimens. I’m intrigued by the dose-dense approach, but before I adopt it routinely, I want to see confirmation from a second trial. Two trials evaluating AC followed by paclitaxel have reported a significant improvement with that adjuvant regimen.
The NSABP-B-28 trial, which added four cycles of paclitaxel to AC, had results similar to the earlier study. Many oncologists have substituted docetaxel for paclitaxel, and the Taxotere-311 data lends support to that in the adjuvant setting. In a younger patient with nodepositive disease who is not eligible for a trial, I am more likely to use AC followed by docetaxel.
The study comparing docetaxel, doxorubicin and cyclophosphamide (TAC) to 5-fluorouracil, doxorubicin and cyclophosphamide (FAC) is a very clean trial.
It is often interpreted as TAC being more effective for patients with one to three positive nodes, but not those with four positive nodes.
That is the way the data were presented, but TAC is pretty effective across the board. Some oncologists have expressed concern about the TAC regimen’s toxicity, and it probably requires the use of growth factors.
— Stephen E Jones, MD
I do not use four cycles of AC. I will consider CMF in some patients with node-negative disease who are at low risk. I usually use six-cycle anthracycline- based regimens — typically FEC. I’m looking forward to the Canadian MA21 trial data directly comparing a six-cycle anthracycline-based regimen to AC followed by paclitaxel. I think this is the “million-dollar question.”
Taxanes clearly offer benefits in the adjuvant setting, and I typically utilize the six-cycle TAC regimen. The disease-free and overall survival of dosedense therapy and TAC are equivalent. Growth factor support, used in conjunction with TAC, reduces the rate of febrile neutropenia to that seen in CALGB-9741.
One of the most interesting questions in adjuvant therapy is: Can a taxane replace an anthracycline? The US Oncology trial presented by Stephen Jones evaluated docetaxel and cyclophosphamide versus doxorubicin/cyclophosphamide. In a population of patients, irrespective of HER2 status, this trial suggests you don’t need anthracyclines. The taxanes may really be usurping the role of the anthracyclines.
The BCIRG adjuvant trastuzumab trial also has a novel, nonanthracycline arm — docetaxel/platinum/trastuzumab — in a HER2-positive population. This combination is based on the preclinical in vitro data of synergism with these agents.
— Denise A Yardley, MD
EDITOR’S COMMENT
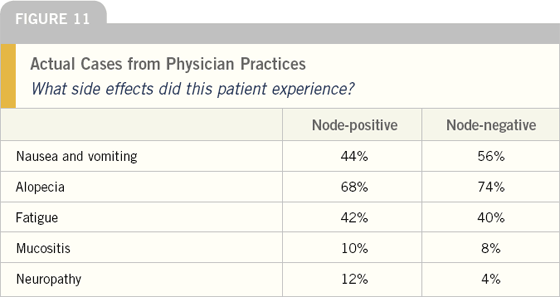
Oncologists reported substantial side effects with chemotherapy. In spite of significant advances in prevention of gastrointestinal side effects, about half of these patients experienced nausea and vomiting. It is also possible that physicians underestimate the side effects patients experience.
Related Comments from Research Leaders
As treatment guidelines continue to evolve with newer data, the relative benefits of treatment in many situations continues to grow. …Parallel to this effort, and of equal importance, is the ability to shield patients that will gain little, if any, benefit from treatment and to identify those who may be at greater risk to suffer from potential toxicities. Until these factors are better defined, the recognition by clinicians of long-term side effects associated with adjuvant therapy is obligatory. Acute toxicities from treatment are often reversible, but late onset adverse effects of therapy can increase morbidity and mortality in long-term survivors, and continue to be of concern. One of the most serious side effects of adjuvant therapy of early breast cancer is cardiac toxicity.
— Theodoulou M, Seidman AD.
Semin Oncol 2003 30(6):730-9.
The side effects of cancer chemotherapy reported by our patients, along with their ranking by relative severity, formed a distinctive profile. The psychosocial complaint affects my family or partner was ranked first as the most severe side effect. Alopecia was ranked second, followed by fatigue (constantly tired). An additional set of psychosocial complaints — effects on work and home duties, effects on social activities, and loss of sexual feeling — ranked fourth, fifth, and sixth, respectively.
— Carelle N et al. Cancer 2002;95:155-63.
Adjuvant therapy decisions are complicated by marginal differences in treatment results and risk-benefit profiles, balancing acute effects with long-term outcomes. Individual patients differ in the value they place on these issues. Retrospective studies report that women may be willing to undergo treatment for as little as a 1 to 2 percent improvement in the probability of survival. Clear communication of benefits and risks is an essential component in enabling as informed a joint treatment decision as possible. Absolute and relative benefits and risks of therapy must be discussed openly.
— National Institutes of Health Consensus
Development Conference Statement
November 1-3, 2000
NSABP-B-30 is an important trial because it will answer whether sequential chemotherapy is better than combination chemotherapy in the adjuvant setting. Patients with node-positive breast cancer are randomly assigned to doxorubicin/cyclophosphamide followed by docetaxel versus doxorubicin/ docetaxel versus docetaxel/doxorubicin/ cyclophosphamide. The rationale for selecting docetaxel is related to the issue of cardiac toxicity. Initial Phase II trials from Europe reported over a 90 percent response rate for paclitaxel when given in combination with doxorubicin. However, an increase in cardiac toxicity was seen when paclitaxel was given in combination with doxorubicin and cyclophosphamide. Although cardiac toxicity may be attenuated by either changing the length of the infusion or by separating paclitaxel from doxorubicin by several hours to a day, these maneuvers may also decrease efficacy.
In Phase II trials, docetaxel did not increase cardiac toxicity when given in combination with doxorubicin. This difference in cardiac toxicity may be related to the different vehicles used to dissolve paclitaxel and docetaxel. Paclitaxel is dissolved in cremophor, which is known to increase doxorubicin’s area under the concentration curve (AUC). Docetaxel, on the other hand, is dissolved in polysorbate, which does not increase doxorubicin’s AUC.
— Eleftherios P Mamounas, MD
No woman with localized breast cancer can know that she definitely will experience a recurrence in the absence of therapy, and even if she did, there is no guarantee that treatment will prevent such a recurrence. Even women with very early stage disease are at some risk of a systemic recurrence after local therapy alone. The potential benefits of adjuvant treatment need to be considered in conjunction with the risk of short-term and long-term side effects. Not only should the patient and physician consider the frequency and intensity of the side effects, but they must also consider how any particular side effect may impact an individual woman’s life.
— Partridge AH et al. J Natl Cancer Inst
Monogr 2001;30:135-42.
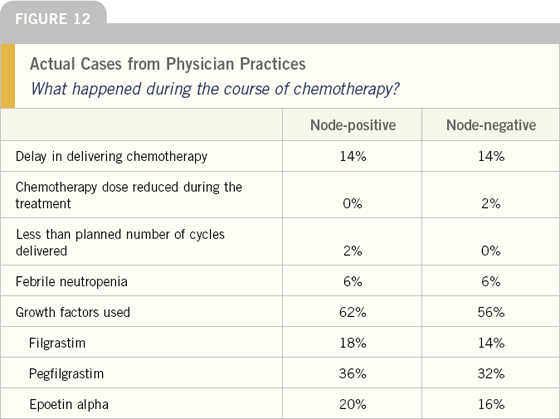
EDITOR’S COMMENT
A recent paper by Gary Lyman — based on data obtained in the late 1990s — suggested that breast cancer patients treated with adjuvant chemotherapy in a community setting frequently have the dose reduced, treatment delayed, or less than the planned number of cycles delivered. Based on the self-reporting of our survey, this trend is reversing. Part of this may relate to the extensive use of hematologic growth factors, which were used in more than half of these patients. Note that pegfilgrastim is used about twice as frequently as filgrastim, undoubtedly reflecting the greater convenience of the longeracting formulation.
Related Comments from Research Leaders
We contracted with over 1,200 nonacademic practices of all sizes (but not academic centers) geographically distributed across the country. We asked them to gather information on their last series of patients receiving adjuvant chemotherapy for breast cancer, starting currently and going backward.
These were patients who were treated with a mixture of chemotherapy regimens from the mid-1990s until early 2000. We are just beginning to evaluate patients treated more recently. Our report published in the Journal of Clinical Oncology focused on approximately six years of data from approximately 20,000 women. The primary area of interest was dose intensity.
It was a very eye-opening experience. We found that the majority of women underwent some degree of reduced dose intensity from the published reference standards. In fact, 56 percent of women across all regimens are receiving less than 85 percent of targeted dose intensity.
In those patients experiencing dose reductions, approximately 40 percent are planned dose reductions, which I believe reflects an intention to “go light” on the first cycle and then raise the doses for subsequent cycles if the patient tolerates therapy well.
That seldom occurs, even in patients who don’t develop neutropenic complications. It’s extremely rare for those cycle-specific dose intensities to be raised during subsequent cycles. Once started low, doses continue to remain low. In the unplanned reductions, we believe 60 to 65 percent are due to physician or patient responses to hematologic toxicities and 40 percent are due to nonhematologic complications.
— Gary H Lyman, MD, MPH, FRCP
In breast cancer, we have data from the Budman-Wood CALGB study, which randomly assigned patients to three different relative dose intensities of CAF. This study was published initially in the New England Journal of Medicine in 1994 and then in the Journal of the National Cancer Institute in 1998 with nine years of follow-up.
A 50 percent reduction in relative dose intensity demonstrated a significant reduction in disease-free and overall survival at five years. The one-third reduction in dose intensity showed a significant decrease in disease-free survival, but overall survival was not yet significantly different.
Why might that be? Can we actually measure the impact of dose intensity and the impact on outcome in patients with a 10 percent, 15 percent, or even 25 percent reduction in relative dose intensity? This is where it becomes very difficult, because it’s largely a power issue. Those studies have not been done prospectively.
In 1995, Bonadonna retrospectively evaluated his CMF data and found enormous differences between women who received more than 85 percent of CMF dose intensity on a 28-day schedule versus those who received less than 85 percent.
Patients who received less than 65 percent of standard dose had a diseasefree and overall survival no different than that of the control group. The problem with Bonadonna’s study is that there are many other potential causes for those reduced dose intensities that might also be related to outcomes.
Retrospective data from the Toronto group and others almost always demonstrated that reducing dose intensity was associated with poorer outcomes, but we really need prospective randomized trials to resolve this issue.
Despite the CALGB trial and a smaller French adjuvant trial — which evaluated FEC 100 versus FEC 50 and showed a significant decrease in disease-free and overall survival with the lower-dose epirubicin — the power calculations would indicate that you literally need thousands of patients in each arm of a trial to measure these kinds of small decrements.
— Gary H Lyman, MD, MPH, FRCP
A retrospective analyses of CMF from Bonadonna in Milan showed that reductions to below 85 percent of planned dose intensity are detrimental to patient outcome, yet there’s interesting evidence from Germany and the United States that oncologists lower and delay dose far more often than anticipated.
Several factors cause dose reduction and delays. To begin, the use of growth factors represents a financial and technical barrier. Also, until recently we didn’t have convincing data that delaying a few days here and there mattered. In addition, a number of toxicities other than myelosuppression, including fatigue, mucositis, diarrhea and nausea, lead to dose reductions and delays.
Every time we dose-reduce or delay, we may be compromising therapy. Clinicians should ask themselves whether they have evidence that this is safe to do. Right now they don’t. All the evidence we have says that dose reductions and delays are not safe.
— Clifford A Hudis, MD
While I rarely use non-dose-dense therapy, for physicians who do and have neutropenic patients on the day of planned therapy, I lean towards the use of growth factors rather than dose-reduction. There is probably a threshold dose important for anthracyclines, and I suspect growth factors are a way of making sure you hit that threshold.
I believe that some physicians “low-ball” patients on the dose of therapy in trying to be “nice” and minimize toxicity. However, if you start at half the dose because you believe the patient is fragile, you’re doing the patient a disservice. I think you need to get the data and treat those patients identically to how they were managed in the protocol.
I think people are becoming more aware that there probably is a threshold effect — the word gets out. I also believe that growth factors allow people to stay on schedule, because you don’t see the profound drops in counts and the high rates of neutropenic fever. Hopefully, as we go on, this will translate to better efficacy outcomes in adjuvant therapy.
I’m still not certain why postmenopausal patients in the Overview appeared to receive half the benefit of chemotherapy of younger patients. Biologically, I can’t understand why that happens, yet it seems consistent over 15 years in the Overview.
I suspect a large part may be dosing issues in the older studies that dominate the Overview. Perhaps this will change in future analyses.
— Hyman Muss, MD
As previously reported, chemotherapy with CMF, as given in our study [Bonadonna], failed to improve outcome significantly in postmenopausal women, particularly those older than 60 years of age. Many oncologists interpreted these results to mean that the predominant effect of chemotherapy was chemical castration.
We have always maintained that the difference in the effectiveness of the regimen between premenopausal and postmenopausal women was mainly, if not exclusively, due to the low dose of chemotherapy that many postmenopausal patients received, either by protocol design or because of protocol violations, including lack of compliance with the regimen for oral cyclophosphamide.
Our results after 20 years of followup confirmed our initial observation. A recent study by the Cancer and Leukemia Group B showed that both premenopausal and postmenopausal women given regimens involving high or moderate doses of cyclophosphamide, doxorubicin, and fluorouracil had significantly better disease-free and overall survival than those given regimens involving low doses.
— Bonadonna G et al. N Engl J Med
1995;332(14):901-6. [Emphasis added,
citations omitted]
Dose and dose intensity of administered chemotherapy are clinically important variables that can be manipulated in an attempt to improve DFS and OS in patients with operable breast cancer.
This trial [Budman-Wood Study] examined these parameters within a conventional dosage range. With additional follow-up since our previous report, we are able to confirm that total dose remains a critical determinant of outcome for this group of patients.
Both the moderate-dose and high-dose arms delivered the same cumulative dose of chemotherapy with no significant difference in outcome (DFS or OS) between these arms for the study as a whole, but significantly better survival than for patients treated with a low-dose- intense arm.
The data therefore suggest that dose reduction, perhaps below a threshold, leads to a relatively worse outcome with the currently available drugs for adjuvant treatment of patients with stage II breast cancer.
— Budman DR et al. J Natl Cancer Inst
1998;90:1205-11. [Emphasis added,
citations omitted]
Next page
Page 1 of 2
|

