| |
HER2 Testing and Trastuzumab |

EDITOR’S COMMENT
Taxanes are the most common agents combined with trastuzumab, and although paclitaxel was the agent utilized in the pivotal trial by Slamon et al, docetaxel is more frequently used by clinicians. Vinorelbine is a common second- and third-line choice.
Related Comments from Research Leaders
All of the chemo-trastuzumab regimens produce excellent response rates between 60 and 70 percent. We’re going to need an adjuvant-like trial to produce sufficient power to prove that one regimen is superior to another.
Outside of the context of a clinical trial, you can take your pick of weekly paclitaxel, weekly docetaxel, gemcitabine, vinorelbine or carboplatin/paclitaxel in combination with trastuzumab. I make the decision on the basis of toxicity after counseling and discussion with the patient.
— Charles L Vogel, MD, FACP, PA
For the time being, trastuzumab should not be given with an anthracycline because of the potential cardiotoxicity. The standard of care is trastuzumab plus paclitaxel, based on the FDA approval. Given the activity of docetaxel in women with metastatic breast cancer and the potential preclinical synergy, there are many physicians who administer trastuzumab plus docetaxel.
When we began studying trastuzumab plus vinorelbine in our first Phase II trial with about 40 women, the combination was well-tolerated and there was an overall response rate of approximately 70 percent. We then conducted a multicenter Phase II trial of trastuzumab and vinorelbine in 55 patients and were again comforted by the safety and efficacy data.
— Eric P Winer, MD
The addition of carboplatin to trastuzumab/paclitaxel in advanced breast cancer improved both the response rate and time to progression. The primary endpoint was the response rate, which improved from 36 percent with the two-drug regimen to 52 percent with the addition of carboplatin, with a p-value of 0.04. We stratified IHC 2+ and 3+ patients, and the response rate in the 3+ patients jumped to 37 percent with the two-drug regimen and to 57 percent with the addition of carboplatin, with a p-value of 0.03. FISH data was collected retrospectively and, although the comparison is not powered for significance, we saw a trend similar to that of the IHC 3+ patients — response rates of 39 percent and 59 percent with the two- and three-drug regimens, respectively.
Time to progression was a secondary endpoint in the trial. The time to progression in the trastuzumab/ paclitaxel control arm was similar to what was seen in the pivotal trial by Slamon and colleagues. The addition of carboplatin increased the time to progression from 6.9 months to 11.2 months. Looking only at the IHC 3+ patients, we saw a similar improvement (7.2 months increased to 13.5 months); similar results were seen in the FISHpositive patients as well.
We looked at survival, although it was early to do so, as over 120 patients are still alive. The preliminary analysis shows a trend for improvement with the three-drug regimen. In the IHC 3+ patients we saw an improvement in survival, with a p-value of 0.06, approaching 0.05, and the FISHpositive population showed a similar trend. It will be important to see if the survival advantage persists.
The trastuzumab/paclitaxel/carboplatin regimen was well tolerated. The only significant difference in toxicity was increased myelosuppression, which we expected to see from adding carboplatin. However, no significant differences were seen in terms of serious complications, such as infectious complications, significant neutropenia or fever. Other toxicities, such as neuropathy, allergic responses, nausea and arthralgias, were comparable in both arms.
— Nicholas J Robert, MD
We’ve been interested in nontoxic chemotherapy regimens and have done a lot of work with vinorelbine and trastuzumab. That combination tends to be well-tolerated, doesn’t cause alopecia or nausea, and I find it appealing for patients who don’t want more aggressive chemotherapy.
— Harold J Burstein, MD, PhD
EDITOR’S COMMENT
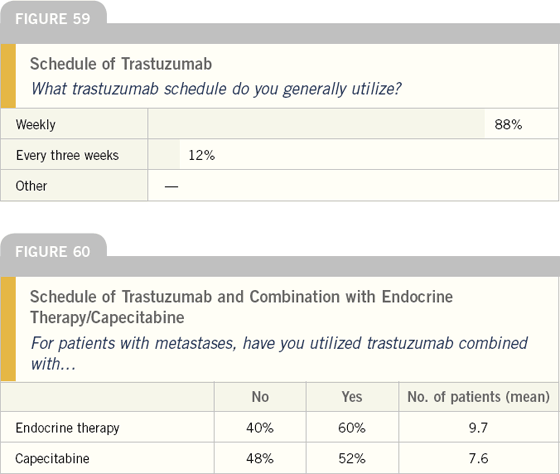
Research from Brian Leyland-Jones has demonstrated the feasibility of using every three-week dosing of trastuzumab, but currently, most clinicians utilize the weekly schedule.
Considerable controversy exists over the role of combining trastuzumab with endocrine therapy. Ongoing clinical trials are evaluating this strategy, particularly the combination of aromatase inhibitors with trastuzumab. A lot of debate has occurred with regard to combining trastuzumab with capecitabine, which does not appear to be synergistic on in vivo testing. Nevertheless, about one-half of oncologists have utilized these combinations in their practices.
Related Comments from Research Leaders
Trastuzumab administered at longer intervals (every three weeks) and at three times the dose is being investigated. Brian Leyland-Jones presented data on paclitaxel with trastuzumab given every three weeks that demonstrated the trough did not go below the desirable level. In fact, the overall area under the curve and the peak concentration are higher without any additional toxicity. This may allow for the convenience of every three-week administration.
I still, however, use weekly trastuzumab. I want a little more toxicity data using it every three weeks. For many drugs, it is the peak level that actually mediates toxicity. That may not be the case with every three-week trastuzumab, but I would like a little longer follow-up, especially for cardiotoxicity.
— Debu Tripathy, MD
We, like many others, have been compelled to switch to triple-dose trastuzumab administered every three weeks. When we discuss Dr Brian Leyland-Jones’ results from his pharmacokinetic studies with the triple-dose, every three-week schedule with our patients, many opt for it, and so far we have not had any problems with that schedule. At this point, however, we really do not have comparative data from large randomized trials. Many of the cooperative group studies evaluating trastuzumab are adopting the every three-week, triple-dose schedule.
In the BCIRG adjuvant trastuzumab trial, trastuzumab will be given following chemotherapy on an every three-week schedule. Over the next couple of years, hundreds of patients will be treated with the every three-week schedule and safety data will be collected. From a theoretical point of view, I am not concerned about efficacy. The peak trastuzumab blood levels are actually higher on the every three-week schedule. Because more trastuzumab is on board, if anything, there could be greater efficacy. I do not believe that will necessarily be the case, but certainly there is no theoretical reason to expect a decrease in efficacy.
— Mark D Pegram, MD
In the subset analysis of the pivotal trastuzumab trial, prior hormonal therapy did not adversely affect the outcomes with chemotherapy and trastuzumab together. For that reason, I feel very comfortable offering endocrine therapy without trastuzumab, as long as it’s clinically indicated, and then bringing in the trastuzumab later.
Many clinical trials are evaluating aromatase inhibitors with or without trastuzumab. Everyone expects to increase the response rate and time to progression by adding the trastuzumab early because it’s an active drug in and of itself.
However, I assume that these studies are never going to answer whether or not there will be a survival benefit because they are relatively small and weren’t designed to have that much follow-up. I think this question will be on the table for a long time.
— Harold J Burstein, MD, PhD
In patients with HER2-positive, ERpositive metastatic breast cancer, I use front-line hormone therapy, assuming they don’t present with life-threatening disease. If the patient responds and then progresses, I continue with endocrine therapy.
If she does not respond initially, then I use trastuzumab monotherapy and add chemotherapy when progression occurs. I haven’t used trastuzumab and hormonal therapy together because I’m unaware of in vitro models showing a synergy between these two therapies.
When using trastuzumab as monotherapy or in combination with chemotherapy, I use the every threeweek schedule. In terms of chemotherapy, I find the weekly carboplatin/ paclitaxel/trastuzumab combination is extremely well-tolerated and active. When a patient presents in visceral crisis, I use either vinorelbine/ trastuzumab or weekly carboplatin/ paclitaxel/trastuzumab.
— Melody A Cobleigh, MD
We compared a weekly schedule to an every three-week schedule of paclitaxel, carboplatin and trastuzumab in patients with HER2-positive metastatic breast cancer. Tolerability was much better for the weekly schedule. Although I thought this would be the case, I was surprised how great the tolerability was for the weekly regimen. Essentially, we observed no significant toxicity, and the activity was very high.
Our trial fits in very well following Nick Robert’s data demonstrating the benefits of adding carboplatin to paclitaxel and trastuzumab, administered once every three weeks.
We presented our results at ASCO 2003. The target accrual for our study was 92 patients, and we will report data on approximately 75 percent of these patients. Because we found the weekly schedule to be better tolerated, after a certain number of patients enrolled, we closed the every three-week arm and continued accrual only to the weekly regimen.
For the weekly schedule, we administered paclitaxel three out of four weeks. I believe it is critically important to take that fourth week off of chemotherapy to really optimize tolerability.
In both arms, we administered the chemotherapy concurrently with trastuzumab for the first six months. Then, at the six-month point, we discontinued the chemotherapy and continued trastuzumab alone — trying to maximize the activity of the interaction of the three drugs while ameliorating long-term toxicities.
— Edith Perez, MD
I still strongly consider hormonal therapy in women with ER-positive, HER2-positive disease; however, evidence suggests that patients with HER2-positive disease may be less likely to respond to hormonal therapy. For that reason, if I were on the fence about using hormonal therapy or moving on to chemotherapy, I would switch to chemotherapy more readily in patients with HER2-overexpressing disease.
When it is time to switch to chemotherapy in patients with HER2-positive disease, most of us believe trastuzumab is the standard of care. The question is whether to use trastuzumab plus chemotherapy or trastuzumab alone. I think in the United States, and certainly in my own practice, trastuzumab plus chemotherapy is more commonly given.
The survival benefit with trastuzumab in the pivotal trial was seen when the combination of chemotherapy and trastuzumab was given up front. Also, there’s a sense that response rates, and therefore control of tumor-related symptoms, are higher when chemotherapy is added to trastuzumab.
— Eric P Winer, MD
If a postmenopausal woman with ERpositive, HER2-positive metastatic disease presents with a minimal tumor burden, I will treat her with an aromatase inhibitor initially and wait to use trastuzumab. I usually start with a nonsteroidal aromatase inhibitor — letrozole or anastrozole — and then move on to exemestane or fulvestrant in patients whose disease progresses.
In patients who need chemotherapy, we use a combination of chemotherapy and trastuzumab because the pivotal trial data demonstrated an improvement in survival for the combination.
When the patients are ready to discontinue chemotherapy, we use the next sequential hormonal agent as maintenance therapy in conjunction with trastuzumab. Studies are currently evaluating the effectiveness of trastuzumab in combination with the aromatase inhibitors, and the results will be very interesting.
— Hope Rugo, MD
I use chemotherapy up front in patients with life-threatening or very bulky HER2-positive disease. In these patients, chemotherapy selection depends on their adjuvant treatment. Traditionally we’ll start with a taxane and trastuzumab. For patients in visceral crisis or with bulky disease.
I’ve been adding weekly carboplatin. Although Nick Robert’s randomized trial evaluated an every threeweek schedule, we see a fair amount of thrombocytopenia with that treatment schedule, so we’ve been using weekly carboplatin, paclitaxel or docetaxel, and trastuzumab. As soon as the patients have a good response, we discontinue the chemotherapy and continue with every three-week trastuzumab alone.
I also use capecitabine with trastuzumab, and it’s been very effective. Patients with HER2-overexpressing disease are often very receptive to capecitabine. So it’s important to use that drug as part of the treatment approach for these patients.
— Hope Rugo, MD

EDITOR’S COMMENT
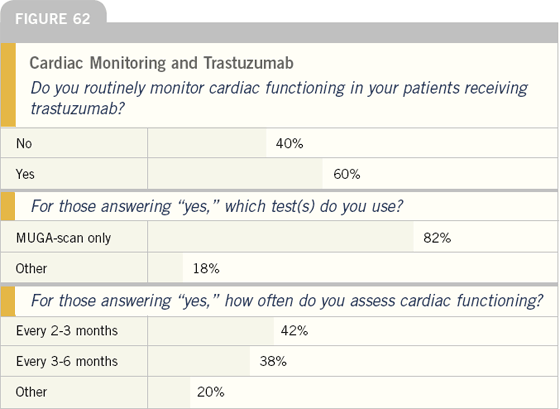
Another controversy in the management of patients with HER2-positive tumors relates to cardiac effects of trastuzumab, and a significant fraction of physicians in practice have had patients with cardiac dysfunction diagnosed either clinically or on screening tests.
Considerable heterogeneity exists regarding screening cardiac testing in patients receiving trastuzumab. About one-half of physicians screen their patients — mainly with MUGA scans.
Related Comments from Research Leaders
When trastuzumab was used in combination with an anthracycline, a significant increase in cardiotoxicity occurred. In the pivotal Phase III trial, about half of the patients with cardiotoxicity had class I and II, and the other half had class III and IV.
Doxorubicin alone is known to cause a nine percent incidence of cardiotoxicity. Patients with clinical cardiotoxicity can be treated with diuretics and ACE inhibitors. When they improve, they can continue on trastuzumab. Or, if the trastuzumab is discontinued, their cardiac function can improve.
We believe the cardiotoxicity associated with paclitaxel/trastuzumab was probably a recall phenomenon because of the data from Chuck Vogel’s study in patients with HER2-positive disease who did not receive chemotherapy.
Those patients were treated with trastuzumab alone, and the cardiac dysfunction rate was just under four percent. All were subclinical. Trastuzumab by itself, in a population of patients with minimal anthracycline exposure, was not a major cardiotoxin.
— Dennis J Slamon, MD, PhD
We evaluated surveillance MUGAs in one of our trastuzumab and vinorelbine trials. We did a baseline MUGA and then a follow-up MUGA at 16 weeks. Among those patients who had preserved left ventricular ejection fraction (LVEF) of 50 percent or greater at 16 weeks, none of them went on to develop symptoms of heart failure or significant declines in LVEF. By contrast, in two of the patients who had declines in LVEF at 16 weeks, we saw problems.
One actually developed heart failure, and the other had a drop in ejection fraction to about 40 percent. While this data only applies to that specific regimen, this has become our routine algorithm.
Anecdotally, I have not seen any late-onset heart failure or changes in LVEF after the first few months of trastuzumab-based therapy. In my experience, cardiac changes usually occur in the first two or three months of therapy, so I think if you recheck the MUGA around three and four months and the patient is clinically stable, you don’t need to frequently check it again.
— Harold J Burstein, MD, PhD
BCIRG-006 is a multinational, randomized, controlled trial for patients with FISH-positive, early stage breast cancer — either node-positive or highrisk, node-negative disease. Patients are randomly assigned to one of three different treatment arms: AC followed by docetaxel, AC followed by docetaxel/ trastuzumab with trastuzumab continued for a total of one year, and trastuzumab/docetaxel with either carboplatin or cisplatin.
For the first time in a large randomized adjuvant study, a nonanthracycline- containing synergistic combination will be put to the test in a very carefully selected patient population. All of the patients must have FISHpositive disease; therefore, I think the trial will define the standard of care for the adjuvant treatment of patients with HER2-positive breast cancer.
The other important component of this trial is safety. A safety data monitoring committee and a specific cardiac safety monitoring committee are monitoring all of the treatment arms in real time, and they have predefined trigger points that call for an interruption in the protocol if any flags for cardiotoxicity occur in the AC followed by trastuzumab/ docetaxel arm.
In fact, the study was designed in such a way that the arm can drop out. If we encounter cardiotoxicity problems, we would still have a two-arm study — one arm with conventional chemotherapy and the other arm with trastuzumab/ platinum/taxane.
It doesn’t appear that cardiac safety is going to be a big issue in the adjuvant trastuzumab trials. Although there was a scare some months ago with the Intergroup trial and one arm was closed temporarily, that arm has reopened and the most recent update, presented by Dr Edith Perez, reveals that the incidence of depressed ejection fractions is the same in all of the arms of the Intergroup trial.
— Mark D Pegram, MD
In January 2002, we received notification of a few patients who developed congestive heart failure on NCCTGN9831. We did not know if it was a real problem or if we just happened to have a few cases at the same time, so we decided to temporarily halt accrual to the third arm of the trial — AC followed by paclitaxel and concurrent trastuzumab — until we had more time to do two things.
First we had to evaluate the clinical course of those few patients who developed congestive heart failure. Second, we had to analyze the data based on all of the more than 700 patients enrolled up to that point. Eventually we found that only a few patients had developed congestive heart failure and that they had prompt improvements of their clinical symptoms with medication.
We submitted this information to our independent data monitoring committee. Because the cases of congestive heart failure were below the threshold we had established in the protocol in June 2002, it was recommended that we reopen accrual to this third arm of the trial. We meet with our cardiologists on a monthly basis to look at all of the data from this study. We have very good compliance with the cardiac testing we recommend as part of this clinical study.
Based on data in the metastatic setting, trastuzumab is associated with congestive heart failure. In the adjuvant setting, it is going to be a matter of assuring that the incidence of congestive heart failure is low and of working on potential predictors of congestive heart failure.
Trials are being devised to address this issue. We are evaluating hypertension, the patient’s age and radiation therapy to the left chest as being predictors of cardiotoxicity. We are also doing quality control to avoid enhancing the potential cardiotoxicity of trastuzumab.
Theoretically, it makes sense that trastuzumab will have a role in the adjuvant setting. However, first we need to finish the clinical trials to prove that point. Then we will have to find ways to ameliorate cardiotoxicity, even if it’s only a few percentage points.
We performed very thorough analyses of ejection fractions as part of NCCTGN9831, and we presented the data at the ASCO 2003 meeting. The specific data are based on the evaluations of ejection fraction after AC chemotherapy.
We have a lot of clinical experience with AC but a scarcity of data regarding its effect on ejection fraction. We found that AC, at a cumulative dose of 240 mg/m2, had a zero incidence of congestive heart failure but decreases in ejection fraction, which tended to be transient.
Our opinion is that ejection fraction may be an interesting marker, but we don’t know if frequent measurements are good in terms of predicting who will develop congestive heart failure.
— Edith Perez, MD

EDITOR’S COMMENT
Clinical research leaders generally do not support the use of trastuzumab as adjuvant therapy outside the context of a clinical trial. The few oncologists who have employed that treatment strategy have done so only in very high-risk cases.
Related Comments from Research Leaders
If someone uses trastuzumab outside of the clinical trial setting, they’re essentially shooting in the dark. We do not yet understand the duration of therapy, the schedule to be used in combination with chemotherapy and the potential risks or benefits the patients may derive.
We have several clinical protocols available. I hope that every woman diagnosed with breast cancer tells her physician, “If I have this bad prognosis, I want to participate in the clinical trial that will help answer the question.”
The NSABP is also conducting a very good trial, also based on solid scientific principles. The NSABP trial has two arms — AC followed by paclitaxel, and AC followed by paclitaxel concurrent with trastuzumab for three months, followed by trastuzumab alone. The NCCTG trial has three arms. NSABPB- 31 is using paclitaxel once every three weeks, as in CALGB-9344, while N9831 is utilizing weekly paclitaxel.
— Edith Perez, MD
In the nonprotocol adjuvant setting, it’s hard to know the right thing to do. I’ve evaluated patients with high-risk disease — 10 or more positive nodes — in whom I’ve considered adjuvant trastuzumab therapy off protocol.
I don’t want to say that this is something that is widely done at our center — it’s infrequent and uncommon. However, the prospects for a patient with that type of disease are really unacceptable.
If you consider that trastuzumab prolongs survival in patients with metastatic disease, biologically there are probably many similarities between high-risk Stage II and advanced disease. Therefore, that would be an interesting patient population to study, and off protocol we have considered such patients for adjuvant trastuzumab therapy.
— Mark D Pegram, MD
Select publications | Previous page
Page 2 of 2
|

