| |
Adjuvant Hormonal Therapy |
EDITOR’S COMMENT
Pathologist Craig Allred has done a series of important studies that suggest perhaps 20 percent of breast cancer patients are misclassified as having ER-negative tumors. The human and public health impact of this finding is enormous.
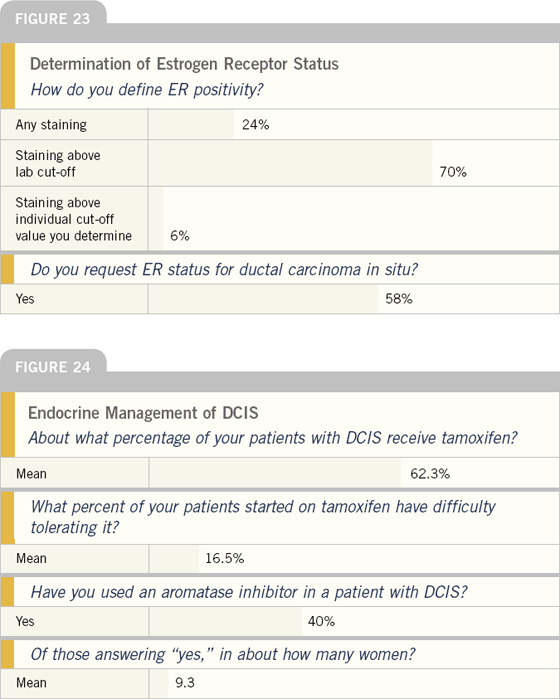
Future surveys will further define how oncologists manage this important question. However, most oncologists accept the qualitative diagnosis of the laboratory doing the assay. More than half consider ER results when deciding on whether to use endocrine intervention for women with DCIS — a follow-up to Dr Allred’s December 2002 presentation to the San Antonio Breast Cancer Symposium.
Tamoxifen is frequently utilized in women with DCIS, and a substantial fraction of these women have difficulty tolerating it. Surprisingly, more than one-third of oncologists have used an aromatase inhibitor in a patient with DCIS. The NSABP is currently conducting clinical trial B-35, which will randomly assign postmenopausal patients to either tamoxifen or anastrozole.
Related Comments from Research Leaders
European studies have shown that approximately 20 percent of ER assays are false negatives when compared to a reference lab. Estrogen receptor testing is not standardized in the United States or Europe, and this leads to a great deal of suboptimal treatment and misunderstanding of breast cancer biology. For years, we thought that some ER-negative patients responded to hormonal therapy; however, I believe this was merely a result of poor assay methodology.
Part of the problem with these assays is technical, and part is in the interpretation. On the technical side, pathologists are just not used to performing immunohistochemistry. The technique is not standardized. Many pathologists come up with their own methods and only do a few cases a week. This lack of standardization and experience causes technical issues and false-negative results. Interpretation of assay results is a problem in terms of both staining and cutoff values. Many laboratories have established a cutoff that is too high and have labeled tumors with ER as being ER-negative.
We have shown in multiple studies in the advanced-disease, adjuvant and DCIS settings that tumors with more than one percent of cells staining positive are hormone responsive, while tumors with less than one percent of cells staining don’t appear to benefit from endocrine therapy.
I believe that medical oncologists often just assume the pathologist is correct. When we started closely reviewing results in our tumor board, it was obvious that there were big problems. Clinicians can insist on having tumors processed in a central laboratory that has a high volume and uses a clinically validated methodology.
— Richard M Elledge, MD
In 1978, the American College of Surgeons conducted a survey demonstrating that 200 out of 24,000 cases of breast cancer were DCIS — less than one percent. The incidence of DCIS exploded in the mammographic era. By screening women, we discovered microcalcifications and other architectural distortions that we otherwise never would have known were present. Some of those women would have developed invasive breast cancer six to 10 years later. Now we intercede in the neoplastic continuum five to 10 years earlier. Today DCIS represents 21 percent of all new cancers.
— Melvin Silverstein, MD
There is variation in defining ER-positivity in Europe and the United States. This variation is extraordinarily disturbing — particularly as our hormonal therapies continue to improve. My feeling is that if there is any receptor present in a tumor, it should be considered positive. Clearly we can miss a very low positive result quite easily, and the result may be that patients who should receive adjuvant endocrine therapy are not receiving it. We need to get this assay correct for every woman.
— Anthony Howell, BSc, MBBS, MSc, FRCP
With minimal training, pathologists in our laboratory were in agreement on discriminating positive from negative tumors in 99 percent of cases. The optimal cut point in our study was a total IHC score of greater than 2, meaning that even patients whose tumors scored 3 (corresponding to as few as one percent to 10 percent weakly positive cells) had a significantly improved response compared to those who had lower scores.
Many hospital and commercial laboratories have converted to assessing ER status exclusively by IHC on archival tissue. They use diverse methodologies, and most have arbitrarily chosen 10 percent or even 20 percent positive tumor cells as their cutoff for defining ER positivity, potentially denying a substantial number of patients the benefits of adjuvant hormone therapy.
— Harvey JM et al. J Clin Oncol 1999;17:1474-81.
Craig Allred presented an abstract at the 2002 San Antonio meeting demonstrating that ER-positive patients in NSABP-B-24 respond well to tamoxifen. The question of whether ER-negative patients respond still seems to be open. ER-negative cases done in Craig’s lab showed no apparent effect from tamoxifen.
In NSABP-B-35, DCIS patients will need ER determinations, and only patients with ER-positive or borderline DCIS can take part in the study. We ask for blocks or core samples at headquarters so that we can do ER determinations in a lab like Craig’s, and an array of other studies to try to understand more about this disease.
— Richard Margolese, MD
Craig Allred reported very provocative data from the NSABP-B-24 trial on estrogen receptor assays in women with DCIS. A central slide review in the NSABP laboratories found that only women with ER- or PR-positive DCIS derived benefit from tamoxifen in preventing ipsilateral breast tumor recurrence and new contralateral primary tumors. They also found a great deal of disparity in reporting the estrogen receptor data, especially in community centers. Based on this data and Dr Allred’s recommendations, it is appropriate to test for estrogen and progesterone receptors in patients with DCIS.
— Hyman Muss, MD
It is clear that DCIS is a highly curable disease from which almost no one should die. If tamoxifen and radiation therapy can reduce the incidence of future invasive cancer to less than two percent, can we achieve even better results?
On the other hand, there are more promising drugs, such as anastrozole. I think it is worthwhile to test anastrozole and see if the small amount of undesired recurrent cancers can be negated. The question becomes: Will anastrozole be any better than tamoxifen, and at what risk?
— Richard Margolese, MD
NSABP-B-35 is the next protocol in a generation of NSABP DCIS trials: B- 17 compared radiotherapy to no treatment, B-24 added tamoxifen to lumpectomy and radiotherapy, and B-35, which opened in January 2003, compares anastrozole to tamoxifen for five years. We’re hoping that anastrozole will be superior to tamoxifen, as it was in the ATAC trial; however, that trial was powered to detect small differences in efficacy.
We debated considerably whether ER positivity should be required for eligibility in B-35. Dr Craig Allred reanalyzed data from NSABP-B-24 and demonstrated benefit from tamoxifen only in those patients with ER-positive DCIS. Ultimately, we decided to limit eligibility for B-35 to patients with ERpositive DCIS. Only a small subset of women with DCIS — approximately 20 percent — is ER-negative. At the current time, I believe it is overly restrictive and authoritarian to dictate that the community standards require estrogen receptor assay prior to treating DCIS.
— Norman Wolmark, MD
NASBP-B-35 is a large study with 3,000 patients, which will go on for the next five years. It is restricted to postmenopausal patients with DCIS who have ER-positive tumors. Studies in the advanced and adjuvant settings found that anastrozole was at least as good as tamoxifen and perhaps superior. Also, the toxicity was less worrisome — anastrozole doesn’t cause uterine cancer or thromboembolism. The issues with anastrozole are that it can’t be used in premenopausal women and it may cause osteoporosis, which can be a serious cause of mortality in elderly women.
— Richard Margolese, MD
EDITOR’S COMMENT
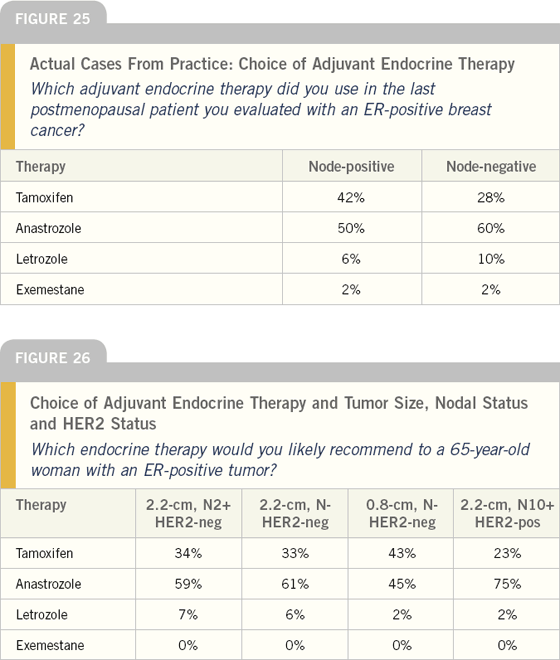
Based on two recent cases in the practice of the surveyed physicians, the most common adjuvant endocrine therapy being utilized is anastrozole. While letrozole is now often utilized in postmenopausal women completing five years of adjuvant tamoxifen, it is not commonly used up front as an initial adjuvant treatment.
We presented a variety of theoretical scenarios in a 65-year-old woman with an ERpositive tumor and found a correlation between baseline risk of recurrence and use of aromatase inhibitors; however, this needs to be further defined in future surveys.
Related Comments from Research Leaders
The results of the ATAC trial are quite compelling. Even if you assume, for the sake of argument, that the curves will come together with further follow-up, the safety profile of anastrozole is still clearly better than that of tamoxifen. I cannot prevent endometrial cancer short of removing the uterus, but I can prevent or treat osteoporosis and fractures. Because the safety profile of anastrozole is better than that of tamoxifen and it is therapeutically superior, I have a problem not offering anastrozole to my postmenopausal patients — not as a neutral choice, but as a better choice. I discuss with my patients the enormous amount of clinical experience we have with tamoxifen, but I would certainly recommend anastrozole as opposed to tamoxifen.
— Gabriel N Hortobagyi, MD
Initially, I had not changed my clinical practice based on the early ATAC results. I was waiting to see more data and whether or not the curves were coming together. However, at 47 months, the divergence of the curves shows a three percent advantage for anastrozole.
There will not be three-percent events in either arm over the next year; therefore, the anastrozole advantage will continue to be the same or greater in the next year. I will now tell patients that there are two options. One option, tamoxifen, seems less efficacious in the short term, but we know its short- and long-term toxicities.
With anastrozole, the time to relapse is substantially improved at the fouryear point, but we really don’t have any long-term safety or efficacy data. There is a risk with either therapy, and many patients will want the new therapy with the potential to be better.
— Peter Ravdin, MD, PhD
We have hard data from the ATAC trial, and we will certainly have even more data this summer after another analysis of the data. Virtually all of the patients will have completed therapy for that analysis.
However, so far the absolute difference between tamoxifen and anastrozole, based on four-year data in postmenopausal women with ER-positive, nodenegative disease, is about one to two percent. If you use Peter Ravdin’s ADJUVANT! program, that’s the one to two percent of adding chemotherapy.
So, I tell patients the story for chemotherapy is not clear-cut, and it’s not clear-cut in patients for whom the benefit is only a few percentage points. Anastrozole may actually accomplish the same thing as tamoxifen and four cycles of AC.
— Gershon Locker, MD

EDITOR’S COMMENT
In a baseline node-positive case, no major difference in selection of endocrine therapy was seen related to age, although some research leaders have noted that they are more likely to use anastrozole in older women because of concerns about deep vein thrombosis and stroke.
When asked to recall how often they prescribed specific aromatase inhibitors as initial adjuvant therapy, oncologists most frequently identified anastrozole, reflecting the fact that the ATAC trial is currently the only study with available data in that setting.
Related Comments from Research Leaders
The biggest change in breast cancer has been the advances in hormonal therapy. I was surprised when the early results from the ATAC trial were reported and the benefits with anastrozole were evident so early.
I think the data from the ATAC trial is very convincing. It is a huge trial with more than 9,000 patients, and it is very unlikely that the curves will change over time. However, I am not sure what the long-term toxicities will be. The data already suggests that there may be a higher risk of fracture in women on aromatase inhibitors.
Tamoxifen is generally a safe drug, but women over the age of 70 have an excess risk of stroke. In women over the age of 70, I am compelled to consider an aromatase inhibitor, mostly because of the risk of stroke.
In premenopausal women with multiple positive nodes, I would consider medical oophorectomy. In those types of patients it might be reasonable to use an aromatase inhibitor. In premenopausal women with multiple positive nodes who stop menstruating after chemotherapy and have low estradiol levels, I would also consider an aromatase inhibitor.
— Debu Tripathy, MD
I do not use aromatase inhibitors other than anastrozole in the adjuvant setting because there are no adjuvant data. While we have to extrapolate in a number of situations, I do not see an advantage for the other aromatase inhibitors from the existing data. It is possible that some time in the future someone will show a distinct advantage to one of these other agents, but at this point, the data were generated with anastrozole, so I use anastrozole.
— Gabriel N Hortobagyi, MD
Counseling postmenopausal patients with regard to adjuvant hormonal therapy requires a lengthy discussion. I refer to studies in the metastatic setting demonstrating a benefit to the aromatase inhibitors over tamoxifen on several endpoints, and I review the ATAC trial results and discuss the risks and benefits of the therapies and the limitations of the data.
Bone density is a big issue for patients. We aim to cure them of their breast cancer, but don’t want to leave them with a second problem. I monitor bone density very closely in patients on aromatase inhibitors. I also counsel patients about the side effects of tamoxifen, including endometrial cancer and thromboembolic events, especially those with comorbid conditions and a propensity for clotting.
Over the last six months, I estimate 30 to 40 percent of my patients have chosen tamoxifen and 60 to 70 percent have chosen an aromatase inhibitor. I believe letrozole and anastrozole are probably equivalent, but I typically use anastrozole because the ATAC data is with anastrozole.
With the recent data on tamoxifen followed by the aromatase inhibitors, this discussion is even more complicated. Some patients are relieved to know some data support changing drugs at the end of five years to give them a little bit more protection.
— Denise A Yardley, MD
In the adjuvant setting I only use anastrozole, because it is the only aromatase inhibitor for which we have data. We can postulate that all three aromatase inhibitors will be active and have similar toxicity, but we don’t know that.
In the metastatic setting, letrozole and anastrozole appear to be very similar in both effectiveness and toxicity. Exemestane has not been very well evaluated, but I would wager that the results will be similar.
In the metastatic setting I don’t have much of a preference for one aromatase inhibitor versus another. Exemestane may have a superior safety profile in terms of bone, but we should think about its potential steroidal effects. We need the adjuvant studies with large numbers of patients to address that issue. We’re not going to get that answer from the metastatic studies, because there have been too few patients.
— Generosa Grana, MD
Bill Miller and Per Lonning warn us not to make assumptions about the efficacy and tolerability of the three aromatase inhibitors because there are very subtle differences between them. We cannot extrapolate from ATAC to exemestane because there may be differences in efficacy and tolerability between the steroidal and nonsteroidal agents. Exemestane is a permanent antiaromatase with weak androgenic effects.
Letrozole and anastrozole are nonsteroidal aromatase inhibitors, but letrozole appears to produce a slightly greater reduction in aromatase. While one might predict this would cause greater efficacy, the tiny trickle of estrogen left by anastrozole may be important for tolerability. We cannot assume a class effect — we must do the trials.
— Michael Baum, ChM, FRCS
I do not use letrozole for adjuvant therapy in the nonprotocol setting. It’s probably equivalent to anastrozole, but I don’t see any significant advantages.
If there was a problem with anastrozole, it would have shown up in this study of 9,000 patients, and I would be able to warn my patients or switch them if necessary. With letrozole, I have no way of knowing if there’s an issue.
I have been looking at whether exemestane might have some advantages compared to anastrozole. There will be trials to test this. Exemestane is a very different aromatase inhibitor — it’s irreversible and it has a steroidal structure.
Early laboratory evidence suggests it will not be associated with bone loss. The resistance mechanisms of exemestane might also be different, which could be better or worse. Remember that tamoxifen can actually be read as an estrogen.
I’m curious to see if a drug with a steroid backbone, such as exemestane, might also be interpreted in some systems as an estrogen. Perhaps the same resistance mechanisms that cause resistance to tamoxifen might also cause resistance to exemestane.
— Peter Ravdin, MD, PhD
As a result of the ATAC trial, my practice pattern changed overnight. I am not treating all of my patients with anastrozole, but I am certainly discussing the results of the ATAC trial and the pros and cons for tamoxifen and anastrozole. I’m using shared decisionmaking with patients to determine which of the agents they prefer.
Generally, I recommend anastrozole; however, there are other factors to consider which would sway me one way or another. Obviously, in women with an absolute or relative contraindication to tamoxifen, it’s a very easy decision. Conversely, there are patients who may have relative contraindications to anastrozole.
The major relative contraindication is severe osteoporosis. The bone mineral density loss associated with the aromatase inhibitors is a concern. Presumably, we can blunt that effect using bisphosphonates, so it is unlikely to be a major problem.
The patient’s nodal status does not make a great deal of difference to me in terms of hormonal therapy recommendations. I look at the patient’s HER2 status and it shades my thinking a bit. Some data exist, although somewhat contradictory, that HER2-overexpressing tumors may be relatively resistant to tamoxifen.
Likewise, data suggest that both letrozole and anastrozole maintain antitumor activity in HER2-overexpressing tumors. I think it would be reasonable to consider anastrozole, in preference to tamoxifen, for patients with tumors that have an IHC score of 2+ or 3+.
— Robert W Carlson, MD
Next page
Page 1 of 2
|

