| |
Editor’s note: Labor and delivery |
Pregnancy and childbirth have always impressed me because of the profound demands placed on the mom-to-be and the highly variable nature of the outcome. The mothers of Einstein, Marcus Garvey and Attila the Hun all followed the same basic pathway and had varying results. So it is with developing a new continuing medical education (CME) program. Conception is the fun part; making it happen is another story. This innocentappearing periodical is actually an experiment in progress that has driven half of our staff insane with our very frequent changes to the prototype made along the way.
Essentially, the idea of Patterns of Care is simple. (Our staff also fought bitterly over the title, with their camp campaigning for “Choices.”) We wish to obtain input from a variety of sources about how patients with cancer are actually managed in community-based practice, and then juxtapose those findings with research leaders’ comments from our CME audio programs and meetings.
This may sound simple, but it is surprisingly challenging to accomplish. The first three issues of this endeavor — forthcoming between now and December — target breast cancer, but we expect and hope to complete future issues about other tumor types. Our approach to gathering data is simple and is one we have been refining for more than 15 years. Essentially, medical oncologists, randomly selected from mailing lists of professional organizations, were asked to participate in a telephone survey in exchange for a modest honorarium. In addition to the usual hypothetical case-based questions, like “How would you treat a patient with…?” we also asked these 150 physicians to describe de-identified cases from their practices in an attempt to better assess their management of patients with breast cancer.
We also asked each physician to describe one of their patients with metastatic breast cancer who had experienced an unusually good response to systemic therapy, and we have included some of these “extraordinary cases” in the Appendix. We often hear about such patients from research leaders and thought it would also be interesting to query community-based practitioners about any similar experiences. Our intent is not to overstate the benefits of systemic therapy, but rather to recognize that while patients with metastatic breast cancer are, perhaps, not “curable,” some may experience responses that are rare or unseen in many other solid tumors.
Our goal for this first pilot survey was to evaluate as many management issues as possible in order to generate hypotheses we could test more definitively in the next two surveys. Therefore, we designed three different tandem questionnaires and divided the 150 medical oncologists into three equal groups. This methodology left us with some substantial statistical variation, but also provided us the opportunity to refine the wording of the questions and the data collection process. In other words, this was a learning experience.
We are also not addressing every possible issue in this complex illness but rather focusing on some of the most controversial and important issues in breast oncology that continually emerge in our CME needs assessment activities.
Most of the results we obtained from these pilot surveys are believable, but some warrant closer study, particularly when inconsistencies are noted. On the other hand, doctors in practice can and do make decisions that perhaps most research leaders would not. Most of these options are evidence-based, but it is curious how uniform research leaders are in their management paradigms — particularly compared to community- based practitioners.
For example, with the oncologist’s permission, I listened to a tape of one of the first anonymous surveys. This very intelligent and well-read physician knew the breast cancer literature inside and out. When asked to describe the last postmenopausal woman in his practice with an ER-positive, node-positive tumor who had recently completed adjuvant chemotherapy, he discussed a very healthy, active woman in her mid fifties with two positive nodes.
Thinking back to our Breast Cancer Update audio program, I would guess at least 90 percent of the research leaders interviewed would use a taxane in such a patient, and three-fourths would use dose-dense AC followed by T. This particular physician had prescribed four cycles of AC. Perhaps he had visualized absolute risk reduction figures in which the added toxicity of a taxane was not worth the incremental benefit. Perhaps he would change his approach to such a patient if he heard 10 research leaders suggest a taxane. Perhaps the 10 researchers would change their minds if they were exposed to this community physician’s thought process about this particular woman. We will address these and other questions in future issues.
The bottom line can be summarized in two words: “Stay tuned.” By the end of the year, we will have a better idea of where things are and maybe where they need to go in breast cancer management. Hopefully, the fruits of this labor will be interesting and provocative.
— Neil Love, MD
NLove@ResearchToPractice.net
EDITOR’S COMMENT
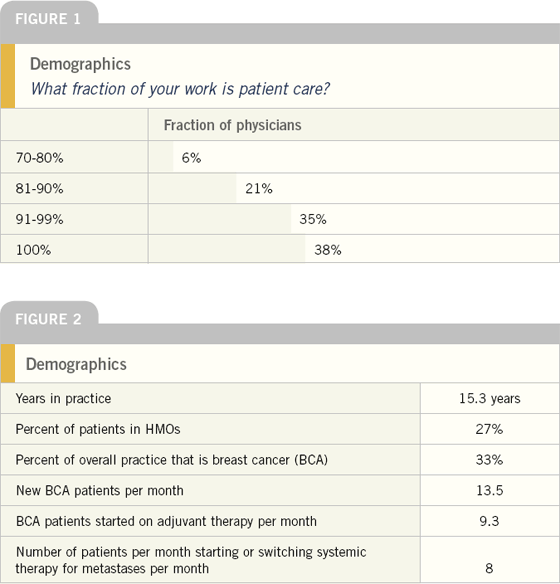
The medical oncologists recruited for this survey were randomly selected from the national mailing list utilized for the Breast Cancer Update audio series. These physicians spend the vast majority of their time in direct patient care.
On average, these physicians have been in practice for 15 years. Approximately one-fourth of their patients have HMO insurance coverage, and about one-third have breast cancer — the largest patient segment in contemporary oncology. These physicians make important breast cancer treatment decisions an average of three to four times a week — both the initiation of adjuvant systemic therapy and starting or switching systemic therapy for metastatic disease.
Related Comments from Research Leaders
One of our major goals is to fully educate our patients by giving them relevant, accurate and complete information so that they understand their prognosis, treatment options and the benefit-to-risk ratio they will face with each of those options — but we can’t stop there.
We also need to make a recommendation after that education. Obviously this recommendation will incorporate our biases and prejudices, but we are better qualified — even with those biases and prejudices — than a patient who just had “Oncology 101” during the previous 20 to 30 minutes. I feel very strongly about that.
Over the past 30 years in medicine, we have moved from a paternalistic approach to the other extreme. Many of my colleagues try to be so neutral that they do not make a recommendation. The burden of decision-making has been removed completely from the physician, who is best qualified to make that choice or recommendation, to the patient, who sometimes is — but most of the times is not — in the best position to make that choice without guidance.
I understand and agree that patients need to have autonomy. We clearly have the obligation to inform them fully, but I think we need to go beyond that. We have to get to know our patients and understand their motivations, their understanding of risks and benefits, their definition of therapeutic gain and their acceptable level of risks and side effects.
As physicians, we need to help them make a decision. To abrogate that responsibility is an unfortunate — and I hope temporary — trend in the medical profession.
— Gabriel N Hortobagyi, MD
Medical treatment decisions in early as well as advanced breast cancer follow established guidelines clearly defined by consensus statements, meta-analyses, and evidence-based insights into the biology of the disease, and treatment efficacy.
Nevertheless, to treat patients “holistically” while remaining mindful of individual characteristics, physicians must consider patients’ concerns regarding the course of their disease, their distress, and, in advanced disease, their knowledge of a limited life expectancy when making treatment choices. Such considerations will contribute to a more satisfactory patient-physician relationship and superior quality of life.
— Zielinski CC.
Semin Oncol 2003;30(2 Suppl 3):27-9.
EDITOR’S COMMENT
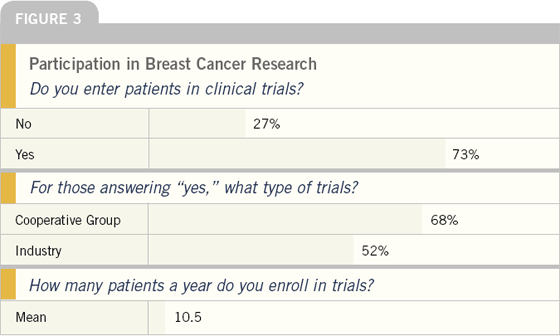
Approximately three-fourths of oncologists enter patients in clinical trials, including both cooperative group and industry studies. Other surveys and keypad polling at meetings have suggested that the most significant impediment to clinical trial participation has been inadequate reimbursement and the amount of time and office support staff required for participation.
Related Comments from Research Leaders
Barriers to clinical trial accrual are multifactorial, and the Cancer Trials Support Unit (CTSU) was designed to attack several of them. Having the infrastructure support — the research nurse support, the IRB support and the financial support — to actually carry out the research is critical when deciding, especially in community practice, to participate in clinical trials. In addition, randomization can be a problem for some physicians and patients. While not able to handle all those issues, the CTSU was designed to reduce the burden of the regulatory paperwork.
For many physicians who choose not to participate in clinical trials, randomization is an issue. We, as physicians, feel that we know the right answer although, time and again, the trials have shown that we don’t know the right answer or that our initial intuition isn’t correct. Many physicians like to go with their bias or intuition and don’t want to randomly assign patients to therapy. In addition, randomization takes more time on the part of the physician. They must explain the pros and cons, as opposed to just presenting a patient with a definitive treatment plan.
It takes a special type of physician who’s willing to put biases aside and take the necessary time to explain why the choice of the therapy will be assigned randomly and why that makes sense in this situation. We always have a harder time when the trial is comparing a treatment to no treatment. The physicians who utilize a particular treatment are biased that the treatment will work.
— Jeffrey Abrams, MD
Arguably one of the most important advances during the last 50 years has been the introduction of prospectively randomized controlled trials to clinical medicine. Such trials provide information about the natural history of a disease and evaluate the worth of a particular therapy. Moreover, they allow for testing of biological hypotheses and, thus, provide a mechanism whereby the scientific method can be applied to clinical problem-solving. By replacing anecdotal information (which has influenced therapeutic decision-making in the past) with more credible and substantive data, clinical trials play a major role in transforming the practice of medicine from an art to a science.
As a vital component of the “research chain,” clinical trials are an essential link between the laboratory and the clinic, providing means for determining whether the use of laboratory findings in the treatment of patients is justified. Without trials, much of the scientific information currently being reported could not be evaluated for its therapeutic worth.
— Bernard Fisher, MD
News from the Commision on Cancer of the
American College of Surgeons 1991;2(2).
The randomised controlled trial has become the gold standard for evidence-based medicine; through the unbiased comparison of competing treatments it is possible to accurately quantify the cost-benefits and harm of individual treatments.
This allows clinicians to offer patients an informed choice and provides the data on which purchasing authorities can make financial decisions. We, of course, subscribe to this view but also recognize this as a gross over-simplification of the power of the randomised controlled trial. The randomised controlled trial is the expression of deductive science in clinical medicine.
Not only is it the most powerful tool we have for subjecting therapeutic hypotheses to the hazard of refutation, but also the biological fallout from such trials should allow clinical scientists to refine biological hypotheses. Trials of treatments for breast cancer have, at least twice, contributed substantially to a paradigm shift in our understanding of the disease.
— Michael Baum, ChM, FRCS;
— Joan Houghton, Bsc
Br Med J 1999;319:568-571.

EDITOR’S COMMENT
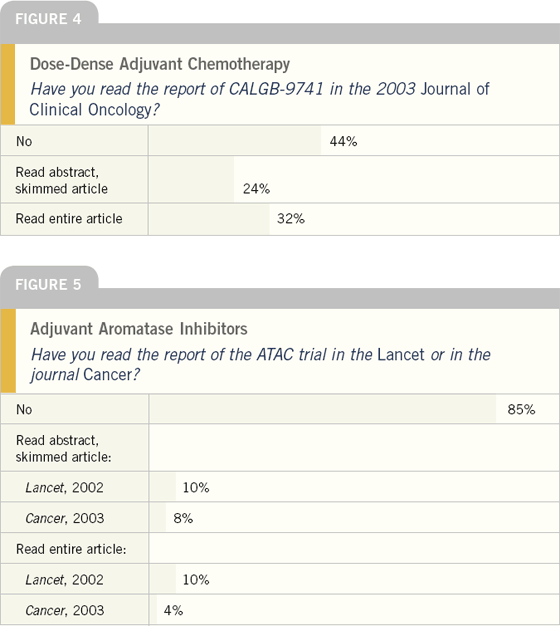
One of the key factors in the extensive utilization of our audio series for oncology healthcare professionals has been the opportunity to “multitask” and listen while driving an automobile or (less frequently) while exercising. The challenge of keeping up to date with increasing demands on physician time is reflected in a finding related to CALGB-9741 — one of the most important clinical research databases reported in oncology in the last few years. Most physicians have stated that they heard about this study — which reported a significant benefit to dose-dense adjuvant chemotherapy — soon after it was presented by Dr Marc Citron in San Antonio in December 2002. However, one year after the formal publication of the paper on this landmark trial, only about one-half of oncologists recalled reading the article, and many had only read the abstract.
The initial results of the ATAC trial were presented by Dr Michael Baum at the December 2002 San Antonio Breast Cancer meeting. This study quickly shifted one of the most important paradigms in adjuvant endocrine therapy, and oncologists promptly heard about this landmark presentation through a variety of sources. However, the majority of oncologists have not read either of the two papers published on these findings. One likely explanation for this is that the journals in which these important papers were published — Lancet and Cancer — are not as widely read by medical oncologists as the Journal of Clinical Oncology.
At the time of the survey, two major papers published in the New England Journal of Medicine reported a benefit for the adjuvant sequence of tamoxifen followed by aromatase inhibitors in postmenopausal patients. One paper, authored by Goss et al, focused on the use of letrozole versus placebo in women who had completed five years of adjuvant tamoxifen. The other study, reported by Coombs et al, demonstrated that women who had received two years of tamoxifen or switched to exemestane faired better than those who continued on tamoxifen. A previous trial reported by an Italian group at the 2003 San Antonio meeting demonstrated that women who had received two to three years of tamoxifen or switched to anastrozole fared better than those who continued on tamoxifen. Oncologists were well aware of this important emerging clinical research data.

EDITOR’S COMMENT
ASCO has developed a set of guidelines regarding the use of tumor markers — such as CA 27.29 and CA 15.3 — suggesting that serial monitoring does not have a reliable research evidence base to justify this management practice. In contrast, many oncologists utilize this approach in both the post-adjuvant and metastatic settings.
Related Comments from Research Leaders
Although I generally agree with the American Society of Clinical Oncology (ASCO) guidelines that we should not be using markers because large studies have shown that they don’t have a benefit, there are times when serial marker measurements may be appropriate. One example might be a patient with inflammatory breast cancer, when you have a suspicion that something is not right.
Another example would be the patient who is at very high risk for relapse. In these situations, we are hoping to diagnose the recurrence before it seriously threatens the patient’s life or compromises a major organ system that would make it difficult to give therapy.
The rationale for seeing people every three to four months early in their disease and at greater intervals later is that early recurrences tend to be the most aggressive, and later recurrences tend to be more indolent. But some patients have such explosive disease that they should be followed more closely, particularly if something just doesn’t add up, although I wouldn’t do so in general.
— Peter Ravdin, MD, PhD
I’d like to speak for the physicians who do use tumor markers despite the ASCO guidelines. I use them routinely. You have to use a good deal of judgment, and there can be much discussion with patients. Such monitoring doesn’t help all patients, but it does help some patients. In a small subset of patients, you pick up disease early and watch them more carefully.
This sometimes allows you to intervene with hormone therapy at a time when they’re relatively asymptomatic. You may avoid situations in which patients get very sick very quickly and will need chemotherapy. It’s very hard to show this value in randomized trials, but I’ve done it routinely over the past 15 years.
— Stephen E Jones, MD
Several randomized studies have evaluated whether a regular schedule of surveillance — imaging studies and tumor marker measurements — would help identify metastatic disease sooner and whether earlier initiation of therapy would improve survival time and quality of life. That strategy failed on both points.
If you do find the metastases earlier and initiate treatment earlier, it isn’t going to extend life. All it’s going to do is force me to tell a patient sooner that she has incurable metastatic disease. I don’t see how that helps in any way, which is why I don’t routinely measure tumor markers after adjuvant therapy.
Patients are often referred to me who’ve been having sequential tumor marker measurements that are rising. The patients undergo every kind of scan that can be done in search of metastatic disease, with tremendous costs and anxiety to the patient and family — and all the scans are negative. The patient is sent to me with the question: “What should we do?” My advice is quite consistent: “Stop checking the tumor markers.”
— Kathy D Miller, MD
Select publications |

