| |
Endocrine Therapy in the Metastatic Setting |
EDITOR’S COMMENT
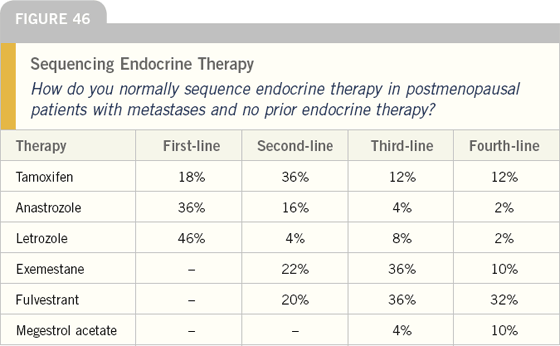
The recent availability of aromatase inhibitors and the estrogen receptor downregulator fulvestrant has complicated the algorithm for management of metastatic breast cancer in postmenopausal women.
A key issue is previous use of adjuvant endocrine intervention, and for patients with no prior treatment, the nonsteroidal aromatase inhibitors are clearly first-line therapy. In this situation, tamoxifen followed by fulvestrant or exemestane are the next agents utilized.
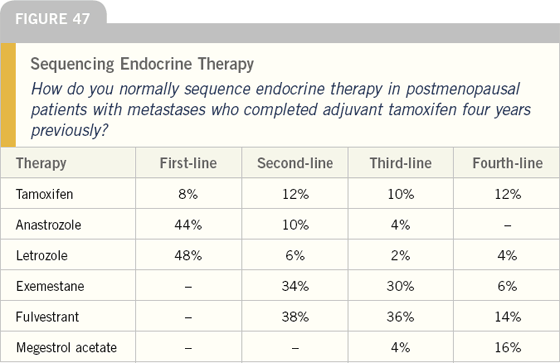
For postmenopausal women who have previously received adjuvant tamoxifen, nonsteroidal aromatase inhibitors are generally first-line therapy, which is then followed by either fulvestrant or exemestane.
Related Comments from Research Leaders
If you look at the data recently published in the Journal of Clinical Oncology in the ER-positive subset, fulvestrant and tamoxifen were basically equivalent. If you evaluate all the patients, fulvestrant demonstrated some numerical inferiority.
One factor you might consider in selecting a hormonal agent is that some patients actually like monthly intramuscular injections as opposed to daily oral therapy. If you asked most oncologists, they would say, “Patients prefer an oral treatment.” However, there is a substantial minority that would prefer to get a shot every month.
— Richard M Elledge, MD
In the first-line study, in the ER/PRpositive group, fulvestrant was slightly (but not significantly) better than anastrozole.
We have to ask, “Why wasn’t fulvestrant better than tamoxifen?” That’s what we expected. The answer may be in the dosing of fulvestrant, because it takes about six months to achieve steadystate levels. Clinical trials will evaluate loading-dose schedules of fulvestrant. Our modeling analyses indicate these approaches will increase the dose of the drug sooner, and then we will be able to investigate whether that is the reason fulvestrant was not better than tamoxifen in the first-line trials.
— Anthony Howell, MD, MSc, FRCP
In a postmenopausal woman whose disease relapses on adjuvant tamoxifen, I use fulvestrant because I’ve seen some very long remissions with it. I will use an aromatase inhibitor later because data indicate that patients with disease that progresses on fulvestrant can still respond to other endocrine treatments (eg, aromatase inhibitors and megestrol acetate).
In this country, fulvestrant is often used as a third- or fourth-line hormonal therapy; however, studies indicate that it might be better than anastrozole following disease progression on tamoxifen. I encourage physicians who are going to try fulvestrant to use it in women progressing on tamoxifen.
— Stephen E Jones, MD
Women with breast cancer who fail on tamoxifen can clearly respond to fulvestrant, and the rate of response is equivalent to that seen with anastrozole. Also, in women with disease that has failed anastrozole who then cross over to fulvestrant, the rate of clinical benefit is substantial and in the range of about 40 percent. Patients who cross over from fulvestrant to aromatase inhibitors also show response rates around 40 percent.
Surprisingly, the magnitude of benefit from fulvestrant does not predict whether the cancer will respond to a subsequent hormonal maneuver. One rule of thumb in the past has been that the magnitude and duration of response to the most recent hormonal therapy predicts for the likelihood of response for subsequent hormonal therapies. A small retrospective study suggests that may not be the case with fulvestrant.
— Robert W Carlson, MD
Fulvestrant 250 milligrams is an effective dose, as demonstrated by the clinical trials. It is as effective as anastrozole as second-line therapy and equivalent to tamoxifen as first-line therapy in postmenopausal women. In premenopausal women, data suggest that 250 milligrams of fulvestrant is not effective at downregulating the estrogen receptor. This raises questions about whether a 250-milligram dose of fulvestrant leads to complete downregulation of the estrogen receptor in postmenopausal women. Could a higher dose of fulvestrant achieve more?
Two strategies exist to increase the dose of fulvestrant. The first is a loading dose sequence. The second is the administration of a higher dose of fulvestrant. For example, instead of administering one five-milliliter injection every month in one buttock, once could use a five-milliliter injection in each buttock, for a total of 500 milligrams. Future studies are needed to determine the dose-response curve for fulvestrant.
— John FR Robertson, MD, FRCS
In patients with hormone receptorpositive disease progressing on tamoxifen, one can switch to an aromatase inhibitor and there’s a good chance the patient will respond. Three commercially available agents have been studied and are approved in this setting, so which agent to use is up to the individual oncologist.
Fulvestrant is also a good choice for these patients. In the two randomized studies comparing it to anastrozole, fulvestrant performed at least as well if not slightly better than anastrozole. Hopefully these patients will benefit from hormonal therapy for an extended period of time, and either fulvestrant followed by an aromatase inhibitor or the other way around will be reasonable alternatives.
— Eric P Winer, MD
For postmenopausal women, a wide choice of endocrine treatment options is available and an optimal sequence has yet to be determined. Options for first-line therapy of metastatic disease include an AI for women who have received adjuvant tamoxifen or tamoxifen for patients who have received adjuvant anastrozole. In addition, data suggest that fulvestrant (‘Faslodex’), a novel estrogen receptor (ER) antagonist that downregulates the ER protein and has no known agonist effects, is a promising therapeutic option that has shown efficacy in the treatment of postmenopausal women with advanced breast cancer. Other agents that may be used in the sequence include the steroidal AI exemestane and the progestin megestrol acetate. The widening range of adjuvant endocrine options therefore represents an opportunity to prolong patient benefits in the treatment of hormone receptor-positive breast cancer, and will require the further refinement of the optimal sequence of endocrine agents for the treatment of recurrent breast cancer.
— Carlson RW, Henderson IC.
Breast Cancer Res Treat 2003;80(Suppl 1):19-26.
In an open-label trial, there were no significant differences between letrozole and anastrozole for the clinical end points of time to progression (primary end point), time to treatment failure, overall survival, clinical benefit, duration of clinical benefit, time to response, duration of response or objective response rate in patients with confirmed hormone receptor-positive tumours. Together these data suggest that once a certain threshold of aromatase inhibition is reached, small differences in oestrogen suppression between the third-generation AIs do not lead to clinically significant differences in overall efficacy.
— Sainsbury R.
Br J Cancer 2004;90:1733-39.
We aim to address whether hormone receptor status influences treatment outcome in postmenopausal women receiving aromatase inhibitors for advanced breast cancer. We include data from three phase III clinical trials, comparing the activity of the new-generation aromatase inhibitors, anastrozole or letrozole, with tamoxifen as a firstline treatment.
For both agents, a significant relationship was observed between hormone receptor status and TTP, with increased TTP seen in patients with a higher confirmed percentage of ERand/ or PR-positive tumors. A relationship between objective response rate (complete or partial response) or clinical benefit (complete or partial response or stabilization for >/=24 weeks) and hormone receptor status was apparent for anastrozole but not letrozole treatment.
— Buzdar AU et al.
Breast J 2004;10(3):211-7.
EDITOR’S COMMENT
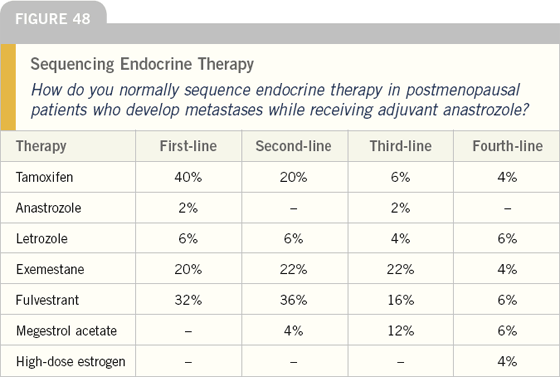
A new generation of patients is emerging who develop metastatic disease while receiving adjuvant nonsteroidal aromatase inhibitors (mainly anastrozole). For these patients, tamoxifen, exemestane and fulvestrant are utilized at first relapse.
Related Comments from Research Leaders
Selection of a hormonal therapy after a patient relapses on anastrozole is a problem. Tamoxifen or fulvestrant could be highly effective, but if the MAP kinase pathway is overdriven from the aromatase inhibition, tamoxifen might act more as an agonist, and fulvestrant might be a better choice.
To my knowledge, in terms of ATAC or other patients who have relapsed on an adjuvant aromatase inhibitor, no data have yet addressed this issue.
— Paul E Goss, MD, PhD, FRCP(CA),
FRCP(UK)
A couple of reports have evaluated the response to fulvestrant in patients who have received an aromatase inhibitor. A fairly small Swiss study reported that about one-third of patients derived clinical benefit from fulvestrant after treatment with tamoxifen or an aromatase inhibitor.
A compassionate-use study, reported at ASCO 2003, reported about 60 patients with fulvestrant as second-, third- or fourth-line therapy. Fulvestrant had more than a 50 percent clinical benefit rate in those patients.
The sequencing paradigm will probably shift because more patients will be treated with adjuvant anastrozole. We don’t know where fulvestrant will fit into that sequence in a patient who has never received tamoxifen and whose disease relapses after adjuvant anastrozole.
— Stephen E Jones, MD
Hormonal agents have a confirmed role in the management of postmenopausal women with receptor-positive advanced breast cancer. Until recently, tamoxifen has been the accepted agent for treating these patients. However, accumulating evidence suggests that the new antiaromatase agents will replace the antiestrogens as the preferable option in hormone-naive patients.
Comparative trials indicate that the aromatase inhibitors, anastrozole and letrozole, and the aromatase inactivator,exemestane, have at least equivalent efficacy to tamoxifen with similar or superior tolerability.
These agents are also more effective than the progestin, megestrol acetate, when studied in patients progressing on tamoxifen. The improved aromatase selectivity and high potency of these antiaromatase agents when compared with earlier agents have resulted in improved efficacy and tolerability.
Additionally, no cross-resistance has been reported between the antiaromatase agents and tamoxifen or, in some instances, among the antiaromatase agents themselves.
— Vogel CL.
Anticancer Drugs 2003;14(4):265-73.
The endocrine cascade for the treatment of premenopausal women with metastatic disease now involves the concurrent or sequential combination of a luteinizing hormone-releasing hormone analogue and tamoxifen, whereas the cascade for the treatment of postmenopausal women can begin with tamoxifen followed by an aromatase inhibitor or with an aromatase inhibitor followed by tamoxifen.
The optimal cascade following the use of an aromatase inhibitor and tamoxifen in postmenopausal women remains unclear, but fulvestrant and megestrol acetate or the use of an aromatase inactivator (exemestane) following an aromatase inhibitor are all available options with some activity. Over the next few years, clinical trials will clarify the optimal sequence of endocrine therapy for postmenopausal women.
— Pritchard KI.
Clin Cancer Res 2003;9(1 Pt 2):460S-7S.
Next page
Page 1 of 2
|

