| |
Endocrine Therapy in the Metastatic Setting |
EDITOR’S COMMENT
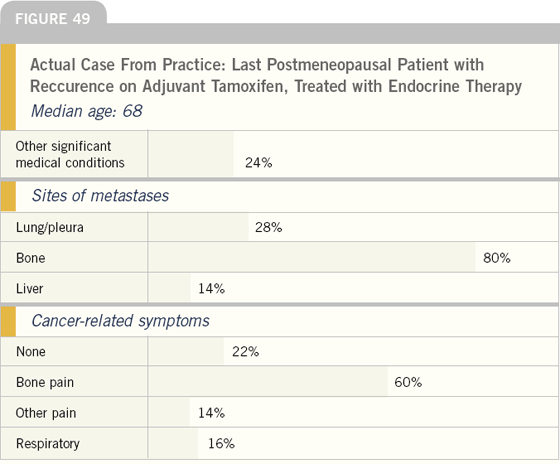
To obtain a more accurate assessment of this important issue, we asked these oncologists to discuss the last woman in their practice who developed disease recurrence while receiving adjuvant tamoxifen. Approximately one-fourth of these women had significant comorbid conditions, and more than three-fourths had tumor-related symptoms.
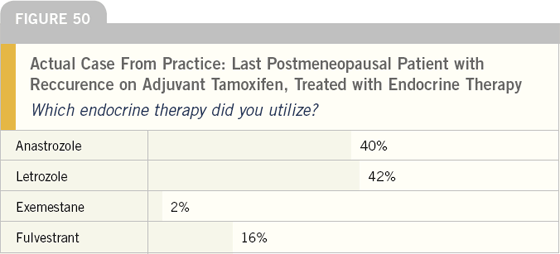
Although randomized clinical trials have demonstrated at least an equivalence of aromatase inhibitors and fulvestrant in this situation, most of these patients were treated with a nonsteroidal aromatase inhibitor. The likely rationale for this choice is avoidance of the intramuscular injection.
Related Comments from Research Leaders
When I see a postmenopausal patient who has relapsed on adjuvant tamoxifen, I tend to use an aromatase inhibitor followed by fulvestrant when the disease progresses. In the frontline metastatic trials of aromatase inhibitors versus tamoxifen, data demonstrate that regardless of whether you administer an aromatase inhibitor after tamoxifen or tamoxifen after an aromatase inhibitor, the result is a 40 to 50 percent clinical benefit rate.
In the second-line setting, if you administer fulvestrant after an aromatase inhibitor or an aromatase inhibitor after fulvestrant, the result is approximately a 33 percent clinical benefit rate. These are all small trials and most of them are not randomized, but they show that any of these regimens can be effective and no mandatory sequence exists for these agents.
— Joyce O’Shaughnessy, MD
The third-generation AIs, anastrozole and letrozole, have been shown to be as effective or more effective than megestrol acetate and tamoxifen as secondand first-line therapies for the treatment of postmenopausal patients with metastatic breast cancer, and exemestane has been approved for second-line use. Fulvestrant has been shown to be as effective as anastrozole as secondline therapy for metastatic breast cancer and has been approved in the US for the treatment of postmenopausal women with hormone-receptor-positive metastatic breast cancer following progression on antiestrogen therapy.
— Buzdar AU.
The Oncologist 2003;8:335-41.
In the metastatic setting, letrozole and anastrozole appear to be very similar in both effectiveness and toxicity. Exemestane has not been very wellevaluated, but I would wager that the results will be similar. In the metastatic setting, I don’t have much of a preference for one aromatase inhibitor versus another. There’s been a lot of speculation that letrozole may lead to some amount of adrenal insufficiency. I’m not sure whether that will be true. Exemestane may have a superior safety profile in terms of bone, but we should think about its potential steroidal effects.
— Genererosa Grana, MD

EDITOR’S COMMENT
Almost all oncologists now have experience prescribing fulvestrant, which is associated with very few side effects or problems tolerating the injection.
Related Comments from Research Leaders
In my clinical experience, fulvestrant is very easy to administer and extremely well-tolerated. My patients have not had any problems with the intramuscular injection. One might assume that a pill is more convenient therapy for a patient than an injection, but that is not necessarily so. Convenience is an individual choice. Some patients would rather receive a shot once a month than take a pill every day. Not only has fulvestrant been exceptionally welltolerated, I’ve seen responses in heavily pretreated patients. Fulvestrant also works after multiple endocrine failures, including tamoxifen and the aromatase inhibitors, even in a third- or fourthline setting. We now have a very welltolerated endocrine agent to add to our armamentarium in the metastatic setting.
— Richard M Elledge, MD
I’ve used a fair amount of fulvestrant, and it’s very well-tolerated. We’ve had some very nice responses to fulvestrant, including one of my patients who was on the original clinical trial of fulvestrant versus anastrozole. She was on fulvestrant for three and a half years and now she’s on anastrozole. The injections have not been an issue for patients, and most women are very grateful that the side-effect profile is close to nil. I think fulvestrant probably crosses the bloodbrain barrier and patients do have hot flashes on it, but in general, they’re quite mild.
— Joyce O’Shaughnessy, MD
This was the first randomized trial to compare the efficacy and tolerability of fulvestrant, the new ER antagonist, with tamoxifen for the treatment of postmenopausal women who have received no prior hormonal or cytotoxic therapy for advanced breast cancer. …
Fulvestrant showed neither superiority nor noninferiority to tamoxifen for the primary efficacy end point of TTP. The almost identical median TTP for fulvestrant and tamoxifen (approximately 8 months) in the subgroup of patients with ER and/or PgR tumors (the group intended for treatment with endocrine therapies) indicates similar efficacy for the two treatments against hormone receptor-positive tumors.
— Howell A et al.
J Clin Oncol 2004;22:1605-13.
Comparative data concerning the efficacy, toxicity, tolerability and cost of AI vs tamoxifen continues to evolve with over 40 000 women slated to be involved in clinical trials. Currently, tamoxifen remains an appropriate choice for adjuvant treatment, and will remain so unless a clear survival advantage emerges for adjuvant AI therapy. However, anastrozole is widely seen as a useful alternative, with particular merit for patients prone to the development of serious tamoxifen side effects. For endocrine therapy naïve advanced disease, several trials have provided evidence that a nonsteroidal AI has replaced tamoxifen as optimal treatment.
— Wong ZW, Ellis MJ.
Br J Cancer 2004;90:20-5.
One of the crucial issues in breast cancer today is how best to integrate the various hormonal therapies. We now have a panoply of hormonal therapies available: antiestrogens, aromatase inhibitors, a pure antiestrogen that knocks out the estrogen receptor and the old progestins.
I suspect we’ll shuffle between these agents once we have a better understanding of cell phenotypes. Then we’ll be able to identify the appropriate hormonal therapy for each patient and tailor our treatment before we see actual clinical resistance.
In the metastatic setting, I generally use an aromatase inhibitor first, then an antiestrogen and then fulvestrant. Unless there’s a contraindication, I begin with aromatase inhibitors because I believe sufficient evidence indicates that they are better than tamoxifen for front-line therapy in metastatic disease. I see approximately a 10 percent incidence of articular complaints with aromatase inhibitors, but I’ve found that switching the structure from a nonsteroidal to a steroidal aromatase inhibitor, or vice versa, seems to diminish those complaints.
— Daniel R Budman, MD, FACP
EDITOR’S COMMENT
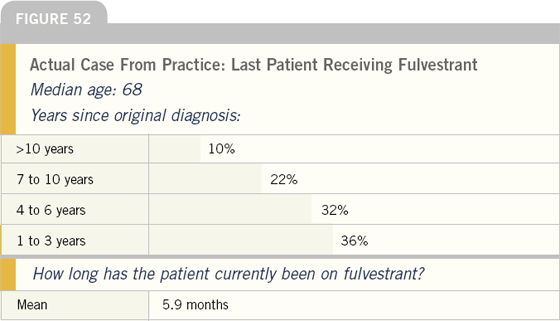
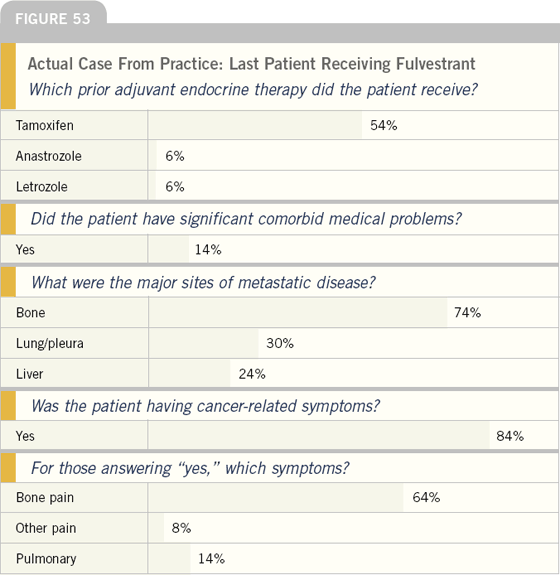
To better assess how fulvestrant is utilized, we asked the physicians to describe the last woman in their practice treated with this agent. On average, patients had been receiving fulvestrant at that point for about six months.
Most of these patients had tumor-related symptoms, and many had received prior adjuvant endocrine therapy.
Related Comments from Research Leaders
Retrospective data suggest that fulvestrant may have a longer duration of response than anastrozole. It’s an interesting finding that would support some of the preclinical models. However, as academic clinicians, we need to be rigorous in our review of the data.
Our group has some patients who have had long durations of response to fulvestrant. One woman was on fulvestrant for more than seven years, another for more than five years and another for four and a half years. We’ve also reported good responders who were treated with other hormonal agents. The only way to test whether patients treated with fulvestrant have longer durations of response is by conducting a randomized trial.
— John FR Robertson, MD, FRCS
Extending the period during which endocrine therapy may be used as an effective and viable treatment option for advanced or metastatic breast cancer in postmenopausal women is an important goal. No curative treatment is currently available for many of these patients, and the ability of endocrine therapy to induce responses without producing debilitating toxicities is very valuable….
This report represents the first examination of sequential endocrine therapy incorporating the ER antagonist fulvestrant before AIs. The results demonstrate that after sequential treatment with tamoxifen and fulvestrant, many patients retain sensitivity to further endocrine therapy with third-generation AIs such as anastrozole and letrozole, or progestins such as megestrol acetate. The rates of CB reported here with endocrine therapy after fulvestrant are similar to those reported for therapy with other endocrine agents (30 to 50 percent).
— Vergote I et al. Breast Cancer Res Treat
2003;79:207-11. [Citations omitted]
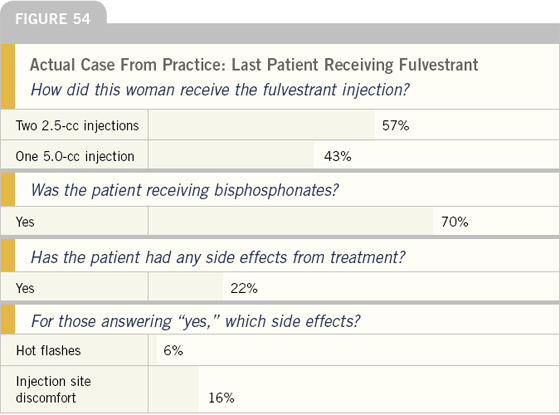
EDITOR’S COMMENT
About half of these patients were treated with a single intramuscular injection of fulvestrant as opposed to split injections, and most patients were also receiving intravenous bisphosphonates and were therefore already committed to a monthly visit to the oncologist’s office. Most of these patients were not experiencing side effects from fulvestrant, but a minority reported discomfort from the injection.
Related Comments from Research Leaders
Injection-site reactions and hot flashes are the only side effects I’ve observed in patients receiving fulvestrant. The administration technique for fulvestrant may affect this infrequently experienced pain. If the injection is inadvertently given subcutaneously into fat, it’s more painful than if it’s given intramuscularly. It may be that many of the women who have pain with the injection are not actually receiving true intramuscular injections; this is more likely to occur in women who are obese.
— Robert W Carlson, MD
It’s great to have fulvestrant as another option for patients who progress following adjuvant tamoxifen, as well as for patients with whom compliance or availability of drugs is an issue. Also, in patients receiving drugs such as pamidronate or zoledronate for bone metastases, the fulvestrant injection can be administered when they come in for treatment so we know they’re receiving adequate care. In terms of tolerability of the injections, I have observed absolutely no problems with them and have received almost no complaints from patients who are receiving the medication. Hot flashes can be difficult to control in many women who have had prior hormone replacement therapy, and I find they’re equivalent whether the patient is taking anastrozole or fulvestrant in the metastatic setting.
— Leroy M Parker, MD
Hormonal therapy (HT) is an important consideration in the management of postmenopausal women with metastatic breast cancer. Despite the fact that the advanced-stage disease is virtually incurable, HTs can offer patients disease control equivalent to that of chemotherapy, but with improved quality of life (QOL)….
Disease that becomes refractory to an initial HT may respond to another agent or class of HTs. Thus, HTs are generally administered sequentially, delaying the need for cytotoxic chemotherapy, which often reduces QOL. Optimal sequencing is thus one of the more important facets of HT. Prior to the release of a number of newer agents, tamoxifen had been considered as initial HT. At present, more agents exist, including the aromatase inhibitors, progestins, and the estrogen receptor antagonist fulvestrant.
— Parker LM.
Clin Ther 2002;24 Suppl C: C43-57.
Following relapse on endocrine therapy for advanced, hormone receptor-positive breast cancer, it is common for patients to experience responses to alternative endocrine agents. Fulvestrant (‘Faslodex’) is a new type of endocrine treatment — an oestrogen receptor (ER) antagonist with no agonist effects. Fulvestrant downregulates cellular levels of the ER resulting in decreased expression of the progesterone receptor. This unique mode of action means that it is important that fulvestrant is placed optimally within the sequence of endocrine therapies to ensure that patients gain maximum benefit. Fulvestrant has shown efficacy when used after progression on tamoxifen or anastrozole in postmenopausal women with advanced breast cancer. After progression on fulvestrant, subsequent endocrine treatments can produce responses in many patients, demonstrating that fulvestrant does not lead to crossresistance with other endocrine therapies. Responses to fulvestrant have also been observed in patients heavily pretreated with prior endocrine therapy. Fulvestrant is a versatile endocrine agent that may be integrated into the therapeutic sequence prior to, or subsequent to, other hormonal therapies…
— Johnston S.
Br J Cancer 2004;90 Suppl 1:S15-8.
Select publications | Previous page
Page 2 of 2
|

