| |
Adjuvant Hormonal Therapy |
EDITOR’S COMMENT
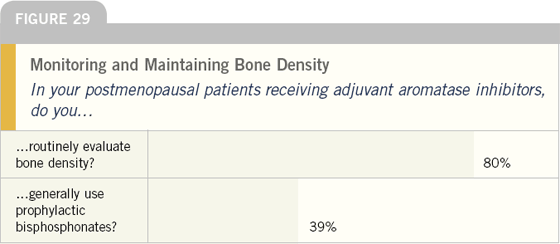
Most breast cancer research leaders believe that a baseline bone mineral density evaluation is indicated when aromatase inhibitors are started as adjuvant therapy, and that bisphosphonates should not be used for prevention of bone loss in women with normal bone density. Oncologists surveyed do not uniformly follow these procedures.
Related Comments from Research Leaders
The early results of ABCSG-12 demonstrate that the combination of goserelin/ anastrozole, and goserelin/tamoxifen to a lesser degree, leads to significant deterioration in bone mineral density in premenopausal women, and that this can be completely counteracted by zoledronic acid.
Even though tamoxifen has an agonistic effect on bone, when combined with the more potent agent goserelin it results in a net reduction in bone density. The bone deterioration is more pronounced with anastrozole/goserelin, but the difference is not significant. The main message is that zoledronic acid was able to completely prevent bone loss, regardless of which hormone combination the patients received.
— Michael Gnant, MD
NSABP-B-34 is evaluating adjuvant clodronate, an oral bisphosphonate, in women with node-negative and nodepositive breast cancer. Data from Germany, Canada and the United Kingdom demonstrate that clodronate reduces bone metastases and improves survival. B-34 will randomly assign women to three years of clodronate or placebo. The choice of adjuvant therapy will be left to the investigator’s discretion. We chose clodronate because it is the only bisphosphonate with data in the adjuvant setting.
If the B-34 results are positive, hopefully clodronate will be FDA-approved. In lieu of the ATAC trial results, it may be reasonable to combine an aromatase inhibitor with a bisphosphonate as adjuvant therapy.
Eventually, the NSABP plans to compare the bisphosphonates to find the best one. It may, however, be difficult — in terms of patient acceptability — to use an intravenous bisphosphonate in the adjuvant setting.
— Eleftherios P Mamounas, MD
Bone loss can be managed. Dr Gnant presented a study at San Antonio looking at ovarian ablation with anastrozole versus tamoxifen and bisphosphonates. They saw protection from bone loss by adding zoledronate. In addition, the women who received tamoxifen and ovarian suppression without a bisphosphonate had a drop in bone loss, which was corrected when they received zoledronate.
The data presented by Dr Gnant is important with regard to anastrozole because without agents like zoledronate, osteoporosis would be a major issue. This study showed that bisphosphonates have the potential to totally prevent the risk of bone loss.
— Peter Ravdin, MD, PhD
Loss of bone mineral density with anastrozole can be monitored. We don’t withhold chemotherapy because we’re worried about white cell count — we give it, but we monitor the white cell count.
Osteopenia is not a dramatic crisis like neutropenia. I would check bone mineral density at diagnosis, upon initiation of anastrozole and annually thereafter. I would intervene with a bisphosphonate if it started to fall. The one adverse effect favoring tamoxifen over anastrozole can be managed.
— Michael Baum, ChM, FRCS
The ABCSG trial 12 demonstrated increased bone density from zoledronate at six months and one year among patients treated with an LHRH agonist plus tamoxifen or anastrozole. We need to follow that study because these were early data from only about 100 patients, and it’s a much larger trial than that.
I’m regularly asked, “Should I automatically administer a bisphosphonate when starting an aromatase inhibitor?” I would prefer to monitor bone density. There are patients who won’t need a bisphosphonate at all. In our update of the MA17 trial of letrozole versus placebo after five years of tamoxifen, we really don’t have substantial numbers of fractures.
Currently, there is a one percent fracture rate in the study. Most of our patients aren’t going to run into big trouble quickly, so you can do a baseline Dexascan™, monitor patients and institute bisphosphonates at an appropriate time based on the WHO criteria for osteoporosis and osteopenia.
— Julie R Gralow, MD
EDITOR’S COMMENT
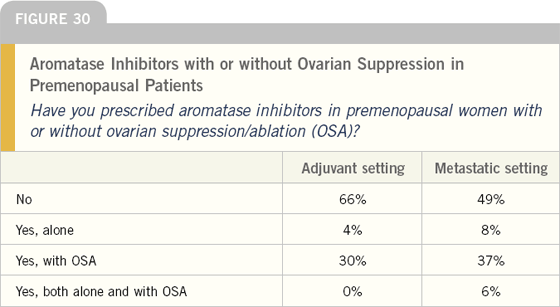
Considerable controversy exists about the nonprotocol role of aromatase inhibitors in premenopausal women. While several important ongoing randomized trials are addressing this crucial question in the adjuvant setting, approximately one-third of oncologists are already adapting this strategy into their practices.
Related Comments from Research Leaders
I’m very enthusiastic about the research strategy of evaluating LHRH agonists with aromatase inhibitors. Extrapolating from the data in postmenopausal breast cancer, which suggested that anastrozole may have superior efficacy compared to tamoxifen, this seems like a rational strategy to transfer to premenopausal women. The two issues are whether or not it is actually going to be efficacious, and what the cost is in terms of side effects. I wouldn’t utilize this strategy outside the context of a clinical trial.
The adjuvant ovarian suppression trial that I am most enthusiastic about is SOFT — Suppression of Ovarian Function Trial. Premenopausal, ERpositive women who may or may not have received chemotherapy will be randomly assigned to tamoxifen for five years, ovarian suppression/ablation plus tamoxifen, or ovarian suppression/ ablation plus an aromatase inhibitor. This very interesting trial will help us address several issues. Does ovarian ablation or suppression add to tamoxifen? And if this is an important strategy, is it better to use tamoxifen or an aromatase inhibitor in women with ovarian suppression?
Two other studies address aromatase inhibitors with ovarian suppression. One is built on the premise — which is pretty popular in Europe — that because we know ovarian suppression is important, some investigators would be unenthusiastic about a trial that didn’t involve ovarian suppression. For those investigators, the trial would be ovarian suppression with tamoxifen or ovarian suppression with an aromatase inhibitor.
The other trial asks the question, “If you do ovarian suppression with either tamoxifen or an aromatase inhibitor, do you really need chemotherapy?” This trial randomizes to chemotherapy or not, plus endocrine therapy.
I believe that will be a tough concept to sell in the United States, but it may have some enthusiasts abroad. I think randomized trials that involve two very different treatments, chemotherapy or not, will be a little more difficult to conceptualize.
These trials were put together by the International Breast Cancer Study Group. They have been looked at by the United States cooperative groups, and different groups will decide whether or not to endorse each trial. Subgroups may decide that they are not as enthusiastic about one design or another, and that they want to put all their effort into one. My personal preference is the SOFT trial, because I think it addresses the issues of interest to many United States investigators.
— Nancy Davidson, MD
The IBCSG is coordinating a series of three nested trials: SOFT, PERCHE and TEXT. These trials address what is probably the most important conceptual question in premenopausal breast cancer right now: Beyond tamoxifen, does planned ovarian suppression benefit patients?
In particular, does it benefit women who receive chemotherapy or women who don’t receive chemotherapy, and if a woman experiences chemotherapyrelated amenorrhea, does she still need ovarian suppression?
We will probably not have the data for at least five or 10 years, but these are very important trials that offer a wonderful opportunity for community oncologists to participate in answering this critical question.
Currently, I consider ovarian suppression for two groups of patients. The first group includes patients at high risk — multiple positive nodes, very highrisk tumors — and particularly young women, less than 35 or 40 years of age, who may not go into menopause with chemotherapy.
The other group includes women who are at the opposite end of the spectrum — very low-risk tumors, smaller tumors, node-negative — for whom the benefits of chemotherapy are very small. I present ovarian suppression as an option for these women, not necessarily in addition to chemotherapy, but perhaps even instead of it.
— Harold J Burstein, MD, PhD

EDITOR’S COMMENT
The Goss paper on post-tamoxifen letrozole had a widespread and immediate impact on practice, and six months later oncologists were uniformly discussing and initiating this therapy. As in the initial adjuvant situation, physicians tend to mirror the source of the research data in choosing an aromatase inhibitor, and letrozole is by far the most common aromatase inhibitor used in this situation.
Related Comments from Research Leaders
Led by the National Cancer Institute of Canada, MA17 randomly assigned over 5,000 postmenopausal women who had received tamoxifen for between four and a half and six years and were free of tumor, to receive letrozole or a placebo.
Letrozole reduced the rate of breast cancer events by about 50 percent, including the risk of distant metastases and the risk of ipsilateral or contralateral breast cancer. The differences were so robust after only two and a half years that the study was closed before completing its planned five-year duration.
The data are exciting because letrozole has the potential to improve the longterm prognosis for the largest demographic group of patients — postmenopausal women with hormone receptorpositive breast cancer. Historically, these women have been offered five years of tamoxifen; now, many such patients should consider taking letrozole after completing that therapy.
It’s always exciting to close a study early because of such good news, but follow-up trials are needed to address unanswered questions about the best way to use letrozole in this setting. There are also concerns regarding the profound estrogen deprivation effects of aromatase inhibitors, particularly osteoporosis. We can study those issues, and interventions do exist, but it means we have to pause before blindly recommending this therapy to everyone.
— Harold J Burstein, MD, PhD
On the basis of these findings, postmenopausal women with hormone-receptorpositive tumors who have completed about five years of adjuvant tamoxifen therapy should be considered for letrozole treatment. However, our results, which necessitated the discontinuation of the study, leave the optimal duration of treatment undefined and the question of long-term toxicity unanswered. Data from other, ongoing aromatase-inhibitor trials will contribute information regarding toxic effects, but the question of the optimal duration of treatment will not be answered by the current trials.
Our study did not address the efficacy of letrozole therapy in women in whom tamoxifen therapy had been discontinued more than three months earlier, but because there was an ongoing reduction in the hazard of recurrence in the letrozole group, the use of the drug in such women should be considered. Consequently, our trial committee has recommended that women in the placebo group in our study discuss their personal risk profile with their oncologist and be considered for letrozole therapy.
— Goss PE et al.
N Engl J Med 2003;349(19):1793-802.
The dramatic results from the NCICCAN- MA17 trial of letrozole after tamoxifen have thrown everyone into turmoil. The levels of significance are so great that neither physicians nor patients can ignore them. However, we don’t have survival data, and it will be difficult to evaluate survival at any point in the future. Additionally, we won’t be able to replicate those results because it wouldn’t be ethical to repeat that study.
It may be reasonable to offer an aromatase inhibitor to patients who completed a five-year course of adjuvant tamoxifen for as long as five or 10 years previously. However, with every year that passes, the absolute risk of recurrence decreases; therefore, the risk-tobenefit ratio changes. Every year the risks become more important relative to the benefit. As the risk of recurrence decreases, the toxicities of therapy become much more important.
— I Craig Henderson, MD
EDITOR’S COMMENT
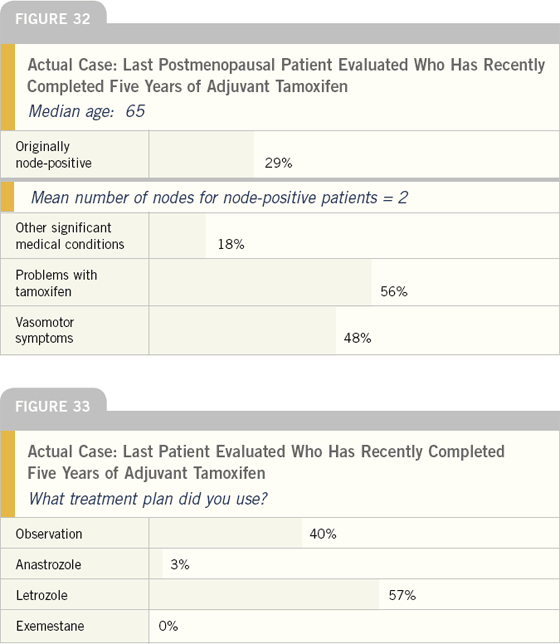
When asked to describe the last postmenopausal woman in their practice who was evaluated after completing five years of adjuvant tamoxifen, a profile emerged of a woman with a node-negative tumor who had significant problems with vasomotor symptoms from tamoxifen.
More than half of these patients were started on letrozole. It will be interesting in future surveys to identify factors associated with prescribing letrozole versus observation in these patients.
Related Comments by Research Leaders
The risk reduction seen in MA17 included both distant metastases and second breast cancer events — either in-breast recurrence or secondary contralateral breast cancers. These local regional recurrences constituted a relatively large fraction of all the breast cancer events seen in MA17. For most women who have had one breast cancer event, their greatest threat to survival is the breast cancer we already know about, rather than a second breast cancer event. For the well-informed patient, the data can be interpreted to offer a secondary benefit — chemoprevention.
The use of aromatase inhibitors in prevention is being explored by a number of investigators. The differences in the ATAC trial are relatively modest, but a trend remains favoring the aromatase inhibitor in terms of preventing second in-breast recurrences or contralateral breast cancers. It suggests that two things are going on — continued control of microscopic distant metastases and ongoing improvement in reducing the risk of primary breast cancer.
—Harold J Burstein, MD, PhD
We don’t know for how long after a patient completes five years of adjuvant tamoxifen it is still beneficial to initiate letrozole. I consider the data from the NSABP-P-1 prevention trial and the patient’s risk to guide me. The P-1 trial showed that if we intervene, we change a woman’s hazard rate for breast cancer occurrence, but we don’t know at what point the reduction in hazard rate becomes so low it is of marginal value.
For a patient with a 1.1-centimeter, node-negative breast tumor, intervention might still be beneficial a couple years after they finish tamoxifen, but for a patient with eight positive nodes and a 2.5-centimeter tumor, I would be willing to treat them further out because the hazard rate is probably still relatively high. When the results of the MA17 trial were revealed, the patients on placebo were offered letrozole even though we didn’t know whether it would be effective two or three years after tamoxifen.
— Clifford A Hudis, MD
All patients with Stage II or Stage III disease who have recently completed a five-year course of adjuvant tamoxifen should receive an aromatase inhibitor. Whether patients with Stage I disease should receive an aromatase inhibitor is an open question because they have a relatively small amount of residual risk. The aromatase inhibitors can be quite expensive for a fairly marginal benefit in patients with very low-risk disease. Additional costs are associated with monitoring bone mineral density or treating with a bisphosphonate. I would like to see more data in Stage I patients.
— Peter Ravdin, MD, PhD
EDITOR’S COMMENT
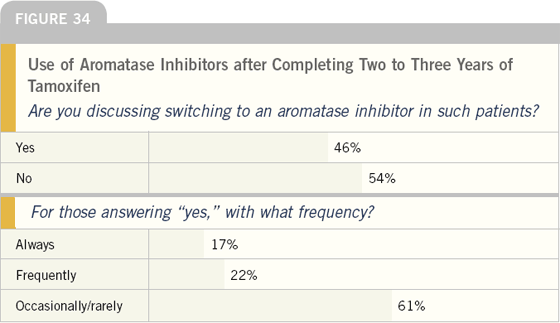
Discussions about switching to aromatase inhibitors occur less frequently with women who have received two to three years of tamoxifen, but almost one-half of oncologists have had these discussions with patients.
Related Comments by Research Leaders
I am usually conservative, especially with my work. This is a relatively small trial and the data are still early, so we need to be cautious and avoid over-interpretation of it. However, that being said, the data speaks for itself and supports an advantage for switching to anastrozole following two to three years of adjuvant tamoxifen. This data also fits in with previous data from a prior study with aminoglutethimide, the ATAC trial and MA17, all pointing in the same direction.
While I do not believe we have the level of evidence necessary to change our guidelines, in my opinion it is reasonable to switch a subset of women from tamoxifen to an aromatase inhibitor after two, three or five years of therapy. There is no reason today to continue tamoxifen in a woman who might be at risk with this drug, because we now have an alternative.
— Francesco Boccardo, MD
Our large, multicenter study challenges the concept of five years of monotherapy with endocrine agents after the surgical treatment of primary breast cancer. Two smaller studies conducted by Italian researchers have used sequential aminoglutethimide after tamoxifen therapy in 308 patients and anastrozole after tamoxifen therapy in 426 patients. Although they were underpowered, both trials suggested that the sequence may be better than tamoxifen alone, supporting the results we present here….
Our results add to the evidence that the sequential use of aromatase inactivators and tamoxifen provides additional options for improving adjuvant endocrine therapy for postmenopausal women with hormone-responsive primary breast cancer. Our results indicate that five years of tamoxifen monotherapy after surgery may be suboptimal for postmenopausal patients with estrogen-receptor-positive breast cancer and suggest that clinicians should consider switching patients to exemestane between two and three years after the start of tamoxifen therapy.
— Coombs C et al. New Eng J Med
2004;350(4):1081-92.
I don’t feel compelled to switch patients to an aromatase inhibitor after just two years of adjuvant tamoxifen. While the switching trials demonstrate the value of adjuvant aromatase inhibitors, we don’t know the ideal time to integrate them. It might be just as good, or better, to wait five years and then switch to letrozole.
On the other hand, with patients just finishing five years of adjuvant tamoxifen, I always discuss the results of MA17. The difference is that switching after just two or three years means trading a proven standard therapy for a therapy that may or may not be superior. After five years of tamoxifen the standard has been to discontinue therapy, but based on the MA17 data, switching to letrozole, which has a demonstrated improvement in diseasefree survival, seems reasonable.
— Clifford A Hudis, MD
In an article published in the Journal of Clinical Oncology in 1996, Dr Saphner et al, reviewing trials from the ECOG database, evaluated the annual hazard rates of recurrence for breast cancer after primary therapy. Patients with four or more positive nodes had a higher risk of recurrence in all time intervals. I believe nodal involvement is key to the risk of recurrence after the first five years.
Letrozole is appropriate in a patient with node-positive breast cancer who completed five years of tamoxifen a year or two ago, but if four or five years have passed and the patient had a small tumor and node-negative disease, the benefit of letrozole would be marginal.
One issue raised by the MA17 and ATAC trials is the selection of endpoints in adjuvant studies. In looking at recurrences, these trials included contralateral, local and regional recurrences. In the future, I suspect we’ll be more interested in the distant disease recurrence endpoint. If we had used that as the endpoint in the MA17 trial, the study would probably still be open and we may have obtained additional information.
— Nicholas J Robert, MD
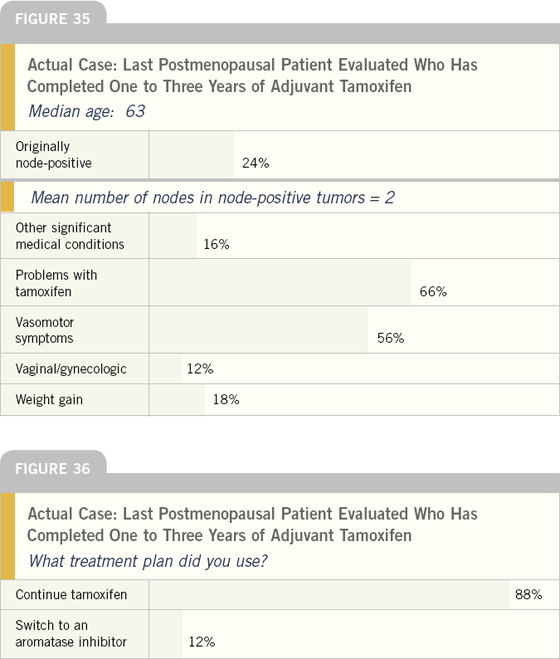
EDITOR’S COMMENT
When asked to describe the most recently treated postmenopausal woman in their practice who was on adjuvant tamoxifen for one to three years, the profile emerged of a woman in her sixties who is having problems with vasomotor symptoms — similar to the profile of the patient after five years of tamoxifen as previously mentioned.
At the time of the survey, oncologists did not frequently switch patients in their first five years of tamoxifen to an aromatase inhibitor.
Related Comments by Research Leaders
Over the past couple of decades, tamoxifen has had a huge impact on the management of breast cancer, but its use in the adjuvant setting may be declining. Several studies have demonstrated the superiority of aromatase inhibitors over tamoxifen, including the ATAC trial, the NCIC-CAN-MA17 trial in which women received letrozole after five years of tamoxifen and two trials in which women were switched to an aromatase inhibitor after two or three years of tamoxifen. The Intergroup study, utilizing exemestane, and Boccardo’s trial, utilizing anastrozole in nodepositive breast cancer, demonstrated an advantage to switching early from tamoxifen to the aromatase inhibitor. When I use endocrine therapy in newly diagnosed patients, I select anastrozole. If I’m going to switch therapy after two or three years of tamoxifen, I use exemestane, but after five years of tamoxifen, I choose letrozole.
— Nicholas J Robert, MD
Select publications | Previous page
Page 2 of 2
|

