|
EDITOR’S COMMENT
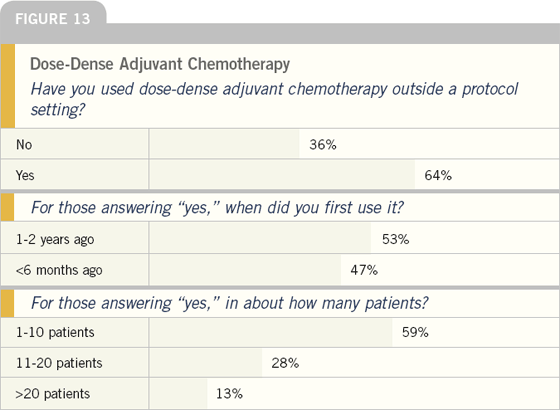
About two-thirds of oncologists have used dose-dense adjuvant chemotherapy, but recently many have only been using it in a relatively small number of patients.
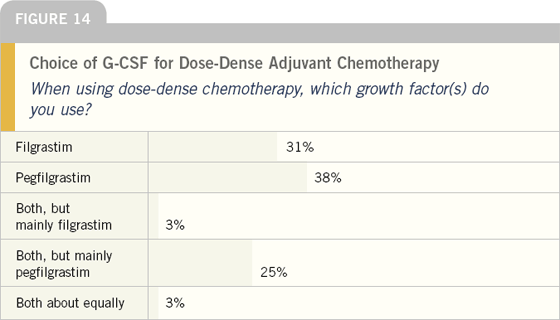
Although the CALGB-9741 trial utilized filgrastim as part of dose-dense chemotherapy, most oncologists are using the longer-acting formulation, pegfilgrastim, for this purpose.
Related Comments from Research Leaders
CALGB-9741 had a two-by-two factorial design, and the results presented at San Antonio compared the two dosedense arms to the two sequential arms. One disadvantage of the two-by-two analysis is that it precludes pair-wise comparison of the two dose-dense arms. However, the dose-dense arms had similar findings.
At a median follow-up of three years, dose-dense treatment was associated with a 26 percent proportional reduction in relapse rate and a 31 percent proportional reduction in mortality. We had expected 515 relapses based on CALGB-8541, the CAF dose-intensive trial; however, there were only 315 recurrences.
The four-year disease-free survival was 82 percent for dose-dense therapy and 75 percent for the every three-week regimens.
I was surprised by the magnitude of the difference — seven percent at four years is significant. We’ll have to see whether the survival benefit is lost or confirmed with further follow-up.
Most patients received the optimal doses of their drugs in all arms. This assured us that the benefits of dose density could not be attributed to a lower dose or further dose delays in the conventional regimens — the arms were balanced in that regard.
The advantages of dose density were not accompanied by an increase in toxicity. In fact, the major difference in side effects was leukopenia, defined as less than 500 granulocytes, which was significantly more common in the every three-week arms, with a p-value of less than 0.0001.
The incidence of hospitalization for febrile neutropenia was also slightly higher in the every three-week arms, but it was uncommon in all arms.
Everyone was concerned about leukemia, but the results do not appear different than the prior protocol, CALGB-9344, at the same exact time point. The incidence is slightly less in the dosedense arms, although not statistically significant. Dose density also appeared to have no impact on cardiac toxicity, which was less than two percent in all arms.
For certain complications, we had information on only the first 100 patients in each arm. One of these was the incidence of red blood cell transfusions, which was 13 percent on the concurrent, dose-dense regimen, while only three percent or less in the other arms.
This is difficult to understand, both from my experience in giving dose-dense therapy and chemotherapy in general, because aggressive use of red-cell stimulating factors generally prevents that complication. This was the only major side effect seen with dose-dense therapy.
Interestingly, severe post-chemotherapy neurologic toxicity was slightly greater in the patients who received concurrent chemotherapy, whether it was every two or every three weeks. I can’t explain that, because we don’t consider cyclophosphamide to be neurotoxic.
It may be just a statistical quirk, but I’ve begun asking my patients on AC if they’re having any neurological problems. Occasionally I hear complaints of paraesthesias, which I had previously attributed to dexamethasone. I’m watching it more carefully now.
Dose-dense therapy is definitely a therapeutic option for patients with high-risk breast cancer at this time. It is not the standard of care, but rather an alternative to discuss with patients at risk for relapse within the next three or four years.
In my older patients who may not be able to tolerate combination treatment, I use sequential dose-dense ATC, and I think we’ll find sequential dose-dense ATC will be tolerated well by the elderly.
I always present patients with their options, and I like to hear what they have to say. In general, patients want the treatment with the most potential for cure. Many also want to receive the treatment quickly — in fact, that’s one of the most common reasons patients express for wanting dose-dense therapy.
Most oncologists like to see five years of follow-up in an adjuvant study. I find when I talk to physicians about emerging trends, I can generally divide the reactions into thirds.
One-third embrace it, a second third are not sure and the remaining third are definitely against it.
I’ve been surprised by how positively dose-dense therapy has been received. As I talk to physicians, I find they are often using or at least considering it. This approach appears to be more widely accepted than I had expected at this time.
— Marc Citron, MD
CALGB-9741 was initially presented by Marc Citron at the 2002 San Antonio Breast Cancer Symposium and subsequently published in the Journal of Clinical Oncology. This Intergroup trial with a two-by-two factorial design asked two presumably unrelated questions.
The first question was whether the concurrent administration of AC was better than the sequential administration. The second question was the comparison of every three-week and every two-week therapy. The every twoweek therapy was administered with granulocyte-colony stimulating factor (G-CSF) support.
Although CALGB-9741 had four different treatment regimens, it was not a four-arm trial; it was two separate two-arm trials with the same patient population. The trial is easily interpretable because the doses of drugs were the same for all patients in all of the treatment arms.
The planned first analysis was not early or preliminary. At this point, the trial had 90 percent power to detect a onethird reduction in the hazard for either main effect — disease-free or overall survival. No significant differences were noted between concurrent and sequential therapy; however, dose-dense therapy (ie, every two-week administration) demonstrated a statistically significant improvement in disease-free and overall survival.
We are confident in these results because the hazard function over time for these arms is significant at all points. The every two-week arms are always better than the every three-week arms, with the margin of benefit appearing to widen.
At four years, the risk of recurrence for the every two-week regimen is 50 percent of the risk of recurrence for the every three-week approach. As with any trial, the issue of toxicity is important for CALGB-9741.
Consistent with CALGB-9344 and in an effort to conserve resources, CALGB-9741 collected safety data for all of the patients only in terms of grade III/IV toxicity and severe adverse events. However, detailed blood count and safety data were not collected.
It was believed that if there were no differences in the detailed safety data from 100 patients per arm, then it did not need to be collected from the whole cohort of 2,000 patients. As a result, detailed safety data is available for only about 100 patients per treatment arm.
Patients treated with G-CSF had a profound reduction in the incidence of neutropenia. Febrile neutropenia was reduced by 50 percent, although statistically one can’t be sure.
We were surprised by the data on red blood cell transfusions. Of the patients treated with concurrent dose-dense AC and paclitaxel, 13 percent received a red blood cell transfusion, which is difficult to explain, because the incidence of grade III/IV anemia was identical in all four arms of the trial. We do not have data about mild (ie, grade II) anemia.
— Clifford A Hudis, MD
EDITOR’S COMMENT
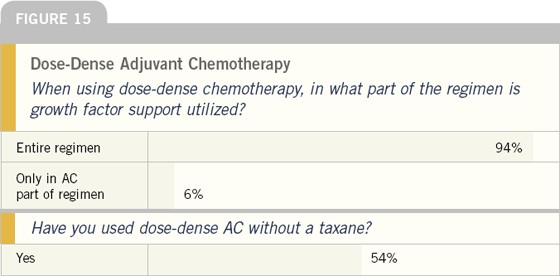
When CALGB-9741 was first reported in December 2002, there was initial speculation that some oncologists would choose to use weekly paclitaxel without growth factor support in the second part of the regimen instead of the dose-dense every two-week approach. Another speculation was that physicians might attempt to use every two-week paclitaxel without growth factors or “as needed” rather than preventively. As evidenced by this survey, neither of these options seems to be commonly utilized in practice.
Related Comments from Research Leaders
Those of us who have been following the developments in the mathematical model really believe that CALGB-9741 is a positive trial because it addresses this mathematical concept. Others are more skeptical and believe the difference is due to the paclitaxel schedule. We don’t have a clear answer.
Ideally, we should evaluate whether the anthracycline or the taxane must be administered in a dose-dense manner. We don’t have time to answer these questions, as too many other important questions need to be addressed. However, I believe it’s reasonable to use dose-dense AC. In Europe it’s not possible to use dose-dense chemotherapy because of financial issues.
— Martine Piccart, MD, PhD
The availability of growth factors and better supportive care measures has enabled us to ask very interesting questions about dose schedule and dose intensity. We have to acknowledge the contributions that Larry Norton and his mathematical models have made in this arena.
CALGB-9741 compared the standard three-week schedule of AC followed by paclitaxel to a dose-dense, every twoweek schedule. The study also evaluated a question of sequential monotherapy versus concurrent therapy. The analyses suggested that, while there was no clinically important difference between sequential therapy and concurrent therapy, the every two-week schedule was superior to the every three-week schedule.
If I give sequential AC and paclitaxel, I give it every two weeks instead of every three weeks, because of the survival advantage associated with that regimen. For node-positive patients, I most frequently use dose-dense AC followed by paclitaxel. We feel quite comfortable with this regimen.
— Harold J Burstein, MD, PhD
I believe in dose-dense therapy because I’ve seen its evolution in the laboratory and the clinic for 25 years, and I believe it has a solid basis. However, no individual can stand up and say this is the new standard of care. We have to see how people are going to utilize this in the community. I would not be shocked to find this approach widely accepted and used, but whether it becomes a new standard of care needs to be defined by the community.
— Larry Norton, MD
As a result of CALGB-9741, the adjuvant trial CALGB-40101 in patients with node-negative disease was amended to use every two-week AC. The proof of greater efficacy with less toxicity was the major consideration in that protocol amendment. In terms of the science, I think it’s reasonable to use every two-week AC without a taxane in a nonprotocol setting. I would hypothesize that patients with negative nodes or a low volume of disease may benefit even more.
From years of trials and the worldwide overview pioneered by Richard Peto, we’ve learned that if something works in patients with node-negative disease, it will work in patients with node-positive disease and vice versa. I don’t think it’s necessary to show that dose-dense therapy is going to work in patients with node-negative disease.
It is a question of the risk of relapse for the patient. A patient with a sixcentimeter, poorly differentiated primary tumor and negative nodes has an enormous risk of relapse, and that patient should benefit as much as a patient with a smaller primary tumor and a few positive nodes.
Aside from any issues of efficacy, the dose-dense approach offers considerable advantages in terms of completing therapy earlier. We offer our patients a choice. We say, “Let’s start dose-dense therapy and see how you do. If you really hate it and you need an extra week, we can always delay things.” I’ve not had a single person who wanted a delay. They just want to complete therapy.
— Larry Norton, MD


EDITOR’S COMMENT
Oncologists seem to be attuned to delivering the planned dose and schedule of adjuvant chemotherapy as evidenced by cases from their practices. When surveyed about their overall use of adjuvant chemotherapy, they report that significant dose reductions or treatment delays are very uncommon, and almost all patients receive the planned number of cycles.
The physicians surveyed were presented with specific patient scenarios related to adjuvant chemotherapy and asked to rank on a zero to 10 analog scale the importance of delivering the planned dose. In these four scenarios, higher scores were given to cases of younger patients at higher risk.
Related Comments from Research Leaders
If changing therapy from every three weeks to every two weeks can reduce the annual odds of death by 31 percent, I shudder to think what going from three to four weeks or from three to five weeks will do in terms of impairing our ability to cure the cancer. Also, with the anthracyclines, the optimal dose seems to be 60 mg/m2 — higher doses don’t provide any benefit, but lower doses rapidly reduce efficacy. I’m worried about both dose modifications and schedule changes.
Reducing doses to avoid toxicities even a little bit gives us, in simulation, a very disappointing result. Cell kill is dramatically impeded by the use of reduced doses of chemotherapeutic drugs.
Consequently, the tumor regrows because the tumor nadir, which may be important for eradication of sublines of tumor cells, is not achieved. To get around dose reductions, the concept of using full doses of drugs sequentially, rather than simultaneously, was hypothesized and tested.
— Larry Norton, MD
Several years ago, data from CALGB- 8541 demonstrated that in the adjuvant setting, full-dose conventional-range therapy was significantly better in the treatment of node-positive breast cancer.
The study examined three cohorts of patients, each receiving different doses of CAF, and evaluated the dose delivery and the total cumulative dose.
Patients receiving the higher doses experienced a marked statistical improvement over the observation period in both disease-free and overall survival in all subsets, and that has continued 10 years later.
There was a steep dose-response curve, so we’ve learned that compromising dose, either initially because of other conditions or reducing dose later, can be detrimental to outcome.
— Daniel R Budman, MD, FACP

EDITOR’S COMMENT
In patients who begin adjuvant chemotherapy without prophylactic growth factor support, neutropenia on the day of treatment is not uncommon. Presented with the scenario of severe neutropenia in this situation, almost all oncologists use the same approach as outlined in most cooperative group clinical protocols — delay treatment until the count recovers, and then include G-CSF with the same dose of chemotherapy.
Related Comments from Research Leaders
Pegfilgrastim is used in the dose-dense arm of the new SWOG-S0221 trial because it certainly makes the regimen more acceptable to patients. Looking at the time course to recovery of neutropenia, it appears that administration every 14 days is possible. Anecdotal results indicate that this is quite tolerable.
Initially, filgrastim will be used in the experimental arm of weekly doxorubicin and daily oral cyclophosphamide. At the University of Washington, pilot studies are being performed to evaluate pegfilgrastim with this regimen. If those studies show that this combination is safe, as expected, then we hope to amend the protocol and use pegfilgrastim in both arms of the study.
— G Thomas Budd, MD
One variable that has changed over time is the use of growth factors. While I can’t overemphasize the limitations of retrospective chart reviews, growth factors are not commonly used early in adjuvant therapy of breast cancer like they might be used in patients with lymphomas or those receiving more intensive regimens.
Approximately one-fourth of patients in our study received a hematopoetic growth factor during the course of treatment, but 85 percent received it secondarily after toxicity occurred. Only two to three percent of patients received primary prophylaxis, and those were probably elderly patients or patients with comorbidities.
— Gary H Lyman, MD, MPH, FRCP
The adjuvant chemotherapy regimens for early-stage breast cancer have not demonstrated an extremely high rate of febrile neutropenia. With AC every 21 days, the rates of febrile neutropenia are quite low but somewhat higher with the addition of a taxane. Severe neutropenia — less than 500 neutrophils at the nadir — is probably more common, although I dare say that many of my colleagues aren’t even looking at it today because the occurrence of febrile neutropenia is so low.
In 1998, Jeff Silber’s group published back-to-back papers in the Journal of Clinical Oncology, in which they developed a model based on retrospective analysis of 100 women receiving adjuvant breast cancer chemotherapy.
They identified three factors in multivariate analyses that were significant predictors of future dose reductions, treatment delays or neutropenic events that would have led them to reduce dose intensity in those patients. These factors were absolute neutrophil count nadir less than 500 in the first cycle, a drop in hemoglobin from baseline to the midcycle of the first cycle and in patients who had previously undergone radiation therapy.
— Gary H Lyman, MD, MPH, FRCP
EDITOR’S COMMENT
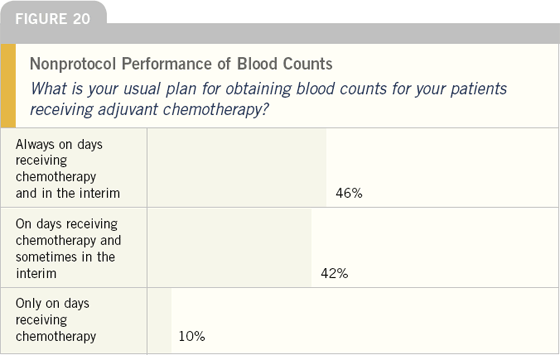
Approximately one-half of oncologists routinely obtain blood counts both on the day of chemotherapy and in the interim. Other oncologists are more selective about interim counts.
In the past, patients with neutropenia on the day of chemotherapy administration were asked to return one week later for a repeat blood count. However, with the increasing awareness of trying to deliver treatment as close as possible to the plan, oncologists now more commonly bring patients back three to five days later for a repeat count.
Related Comments from Research Leaders
Managing patients who present with afebrile neutropenia is a challenge. A key issue is the threshold neutrophil count at which one feels comfortable treating. In my career, I’ve gravitated to using from 800 to 1,000 neutrophils as my cutoff for either delaying or reducing dose.
Typically, I will delay treatment one to three days and repeat counts. I won’t delay a full week, which has historically been the “knee-jerk” reaction. Doxorubicin plus cyclophosphamide has a very abbreviated period of neutropenia. It can go quite low, but usually it’s not very prolonged, which is probably why these women don’t have a very high risk of febrile neutropenia.
In patients with high-risk disease, I do everything possible to avoid reducing their dose. Use of growth factors is an option. Another rational option is to forge ahead with therapy, especially with dose-dense therapy in which we’re automatically using growth factor support. I think we’re going to find that even women in the 21-day cycles are going to receive growth factors for the future cycles, and they’ll probably do fine.
I don’t use growth factors universally. I consider age and comorbidities, and if I think chemotherapy presents a real risk of future complications to the woman, I’ll add growth factors.
In my experience, probably 25 to 30 percent of patients receive growth factor support at some point. I believe the more rational approach is to target it to the group of patients at the highest risk and do it preemptively as opposed to waiting until they’re hospitalized or already neutropenic. Growth factors are much less effective once the patient is neutropenic.
— Gary H Lyman, MD, MPH, FRCP
The current American Society of Clinical Oncology guidelines recommend primary prophylaxis with the CSFs in patients receiving chemotherapy regimens that produce febrile neutropenia in 40% or more of those treated. This recommendation was supported by an early economic analysis based on a decision model incorporating hospital cost data to determine the risk threshold for the cost-saving use of the CSFs.
Updating this model to include current estimates of hospitalization costs, indirect costs such as productivity losses, and out-of-pocket patient expenses reduced the risk threshold for cost savings with CSF to 18%. Recent efforts have focused on identifying individual patient characteristics that might be used to target prophylactic CSF in patients who are at greatest risk.
Should such factors be identified and validated, assessing each patient’s individual risk for neutropenic complications may prove to be a better strategy for the cost-effective use of the CSFs than the risk threshold approach.
— Lyman GH.
Semin Onc 2003;30(Suppl 13):10-7.
Chemotherapy prolongs survival in patients with early-stage breast cancer, and maintaining the chemotherapy dose intensity is crucial for increasing overall survival. Many patients are, however, treated with less than the standard dose intensity because of neutropenia and its complications. Prophylactic colonystimulating factor (CSF) reduces the incidence and duration of neutropenia, facilitating the delivery of the planned chemotherapy doses. Targeting CSF to only at-risk patients is cost-effective, and predictive models are being investigated and developed to make it possible for clinicians to identify patients who are at highest risk for neutropenic complications.
— Dang CT et al. Oncology (Huntingt)
2003;17(11 Suppl 11):14-20.
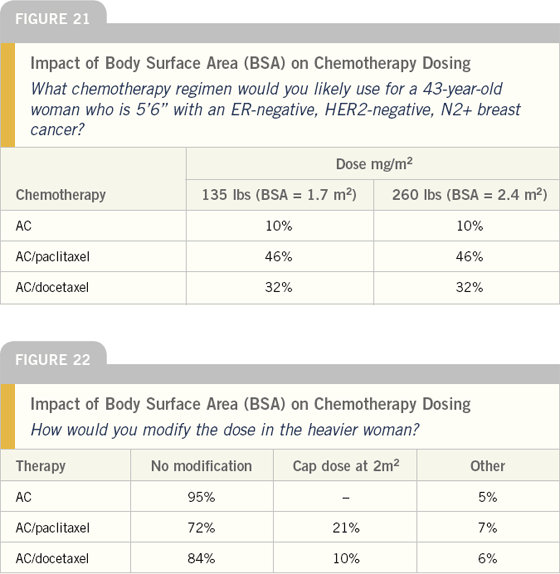
EDITOR’S COMMENT
One of the most challenging questions in adjuvant chemotherapy for early breast cancer is dosing of patients with significant obesity. We presented such a case in our survey, and the regimen selected by most physicians was identical to that selected for the same patient without obesity.
The dosing of the obese patient in our scenario requires further testing in future surveys. Our preliminary findings suggest that most oncologists do not adjust the dose, although a significant minority “round off” to 2.0 m2.
Related Comments from Research Leaders
We have dosed patients by “per meter squared (m2)” since the 1960s, but this was based on the erroneous belief that utilizing body surface area (BSA) normalized dosing — the larger the person, the larger the dose. We now know that BSA has very little meaning; the important considerations are how the individual absorbs or metabolizes the drug, and metabolism varies tremendously.
Obese patients raise particular concern. The CALGB examined this issue across adjuvant treatments and recommended we not dose-reduce even the morbidly obese patient. I must admit, most of my morbidly obese patients have comorbid conditions, such as hypertension, diabetes, et cetera, and I am reticent to administer large doses of cytotoxic drugs to such patients. So, despite the CALGB’s recommendation, I cap the body surface area at 2.0 m2.
— Daniel R Budman, MD, FACP
Recent studies suggest that using body surface area to dose drugs like the taxanes may not be optimal. Some institutions are using “flat dosing” for paclitaxel. It may not be any better or worse to use a flat dose of 300 milligrams. BSA may be a poor surrogate for the metabolic rate. Individual variations are far greater than the BSA variations— it’s an interesting dilemma.
— Clifford A Hudis, MD
BSA was introduced into medical oncology to safely predict a suitable starting dose in Phase I clinical trials from preclinical animal toxicology data. From that starting point in Phase I trials it has spread throughout the practice of oncology with little justification. The formula to calculate body surface area takes two precisely quantifiable variables, height and weight, and estimates a value for surface area. The formula used to do this has never been adequately validated. Very few of the organ functions that determine the pharmacokinetics of a drug are related to body surface area; further when organ function has been related to body surface area other measures such as lean body weight have been found superior to surface area. For the majority of drugs, the relationship between BSA and kinetics has not been studied and where the relationship between BSA and kinetics has been examined only a few drugs such as the taxanes have relationships been found.
— Sawyer M, Ratain MJ
Invest New Drugs 2001
Every oncologist has a threshold at which they become anxious and begin to adjust weight to ideal, or to some compromise between ideal and actual body weight. In our study, that threshold was extremely variable and particularly dramatic above 2.0 m2 body surface area. Many practices have patients with BSAs exceeding 2.75 m2, or even 3.0 m2, and calculating dose based on actual weight can arouse anxiety. However, for patients in whom dose was based on actual body weight, there was no greater hematologic toxicity or later dose reduction or treatment delay, at least not in patients with BSAs between 2.0 m2 and 2.3 m2.
My oncology group is excited about these findings and about trying to reevaluate the early data that served as the basis for our approach of dosing based on BSA. In reviewing those early studies, one realizes that a handful of patients were studied with techniques that we could probably improve upon today. We’re going back and trying to redo many of the early pharmacokinetic studies to determine if basing dose on BSA — of all the possible options that are out there — still seems to be the most rational approach. It seems to be how most physicians are currently calculating dose.
— Gary H Lyman, MD, MPH, FRCP
Select publications | Previous page
Page 2 of 2
|

