| |
Choice and Use of Taxanes |

EDITOR’S COMMENT
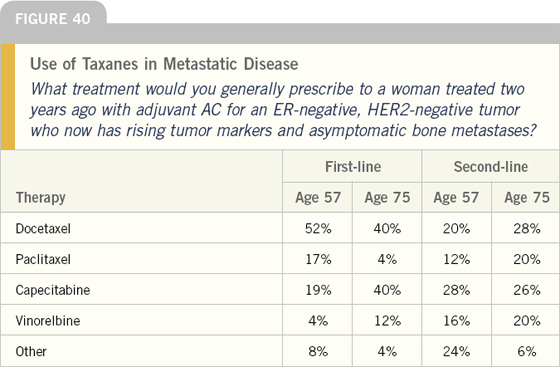
Most women receiving first-line treatment for metastatic disease have had prior adjuvant therapy, and many have at least had AC. Docetaxel is the most common single agent utilized when a patient has previously had AC. Paclitaxel and capecitabine are also frequently selected in this situation. For older women, capecitabine is much more frequently used.
While the strategy of using sequential single-agent chemotherapy is standard in patients who are clinically stable, combination therapy is used by many oncologists in patients with poor performance status. This trend is far less evident in older women.
Related Comments from Research Leaders
The results of the Taxotere-311 trial were surprising, and I didn’t think they would be quite so dramatic. For the evaluable patients, a significant difference was seen in the response rate, time to tumor progression and survival in favor of docetaxel. More toxicity was associated with docetaxel than with paclitaxel, but it was the usual manageable toxicity.
This study basically confirmed that docetaxel was probably a more potent taxane, at least on an every three-week schedule. The survival advantage was surprising because few regimens have a documented survival advantage in patients with metastatic breast cancer.
— Stephen E Jones, MD
Combination versus single-agent chemotherapy in the metastatic setting is currently a big debate in oncology. I use combinations in some patients and single agents in others, and I believe the heterogeneity of the disease warrants that. Dr Sledge’s trial demonstrated the response rate and the time to progression were significantly in favor of the combination regimen, but overall survival was equal to that of single agents with the crossover.
I may consider using a combination regimen to control the disease more quickly in very young patients, those with a very short disease-free interval, visceral disease or a large tumor burden. In the chemotherapy-naïve patient, I typically incorporate a taxane up front either as a single agent or in combination — often with a platinum.
Sequencing of single agents in the metastatic setting is basically a patientphysician decision. I evaluate prior adjuvant therapy, the disease location, the patient’s last regimen, quality-of-life issues and side-effect profiles. I don’t believe we have data suggesting a certain sequence to which one should adhere.
The drug that’s given earliest tends to have the highest response rate, and it drops sequentially thereafter.
— Denise A Yardley, MD
George Sledge’s Phase III trial of singleagent doxorubicin, paclitaxel versus the combination of doxorubicin/paclitaxel as front-line chemotherapy for metastatic breast cancer failed to show a survival benefit for the combination. It’s difficult to demonstrate a survival advantage in front-line metastatic disease because, according to the MD Anderson series, these patients live an average of four years.
What you do early in their treatment may never be reflected in a survival advantage because they have many other opportunities for treatment down the line.
In chemotherapy-naïve patients with metastatic disease, I generally use capecitabine/docetaxel (XT). There’s no evidence that administering an anthracycline after a taxane harms the patient in any way. I eventually use an anthracycline; I just don’t feel compelled to use it up front.
The decision to use single-agent taxane or single-agent capecitabine or the combination for frontline therapy depends on factors such as the patient’s presentation and the extent of her disease. As we begin later-line therapy, when patients become more symptomatic and heavily tumor-burdened, and their life expectancy is shortening, a very reasonable argument can be made for better palliation and maybe even better survival with a well-tolerated combination regimen.
— Joyce O’Shaughnessy, MD
Outside of a clinical trial, a woman who has received an anthracycline as adjuvant therapy could potentially receive docetaxel, paclitaxel, capecitabine or vinorelbine as first-line therapy for metastatic disease. In my opinion, the response rates for these agents are fairly similar. Some oncologists believe docetaxel is the most active agent, but I am not convinced that any of these agents have different activity. I tailor the treatment to the woman and base my decision on the types of side effects the woman would prefer to avoid.
Regarding toxicity, the best agents are probably capecitabine and vinorelbine. Alopecia is often an issue for women, and capecitabine is not associated with hair loss. If one is careful with the capecitabine dose, most side effects can be avoided. Over time, some women may experience chronic changes in their hands and feet, but that is the predominant toxicity encountered with capecitabine.
In addition, I find when it’s time for a patient to switch from hormonal therapy to chemotherapy, switching to capecitabine is not such a big step for them psychologically.
— Eric P Winer, MD
ECOG-1193 compared doxorubicin followed by paclitaxel, paclitaxel followed by doxorubicin, and the combination of the two agents at initial relapse. The overall response rate for the combination of agents was better than that of either single agent. The time to treatment failure was approximately two months longer for the combination than for either single agent, but overall survival and quality of life were identical among the three arms.
My personal bias is this data provided support for the use of sequential singleagent chemotherapy. In my clinic, I find single agents to be less toxic in many cases, and I frequently offer patients with metastatic disease single-agent chemotherapy.
Joyce O’Shaughnessy’s trial demonstrated a survival advantage of approximately three months for the addition of capecitabine to docetaxel in the metastatic setting for anthracyclinerefractory patients. This was a wellconducted, statistically rigorous trial, and the results are certainly believable.
Capecitabine provides a real benefit for patients with metastatic breast cancer, but I don’t conclude that combination therapy is superior to sequential singleagent therapy, and this trial did not test that hypothesis. There was no crossover arm from docetaxel to capecitabine or from capecitabine to docetaxel. In most cases, patients did not cross over to capecitabine. This trial is not comparable to ECOG-1193, which specifically addressed that question.
— George W Sledge, MD
The big question associated with the sequential single-agent versus combination chemotherapy trials is the effect of crossover therapy. In Joyce O’Shaughnessy’s trial, we don’t know what the effect on survival would have been if 60 or 70 percent of the patients treated with single-agent docetaxel were then treated with capecitabine. Maybe we would not have seen a survival difference. Hence, the effect of crossover therapy remains a question in all of these trials comparing doublets to single-agent regimens.
I generally prefer single-agent chemotherapy, but I discuss combination chemotherapy with my patients and offer them a choice. In clinical practice, my approach has been to use combination chemotherapy when I can’t wait for a response.
In the patient with limited disease who needs chemotherapy, in whom I’m hoping to obtain a complete remission, consolidate the sites of disease with radiation or if there is a chance for a prolonged remission, I would probably also favor combination chemotherapy.
If the treatment is strictly for palliation or to try to control the cancer, I’m probably going to use sequential singleagent chemotherapy.
— Stephen E Jones, MD
EDITOR’S COMMENT
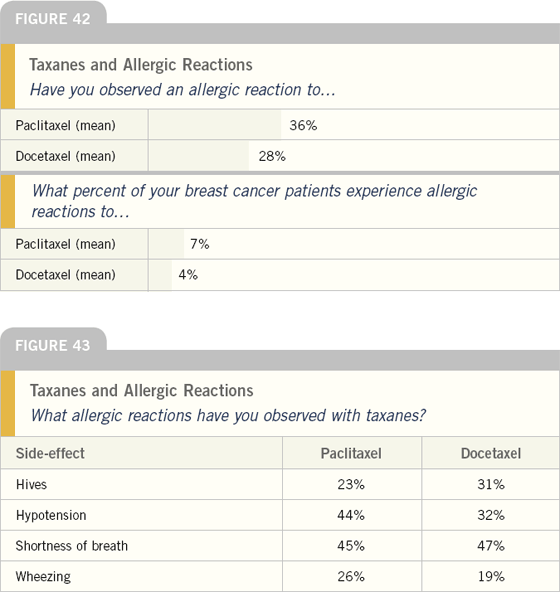
Approximately one-third of oncologists have observed allergic reactions to either docetaxel or paclitaxel, but this only occurs in a small fraction of patients. The spectrum of allergic symptoms observed in patients receiving paclitaxel is very similar to those observed in patients receiving docetaxel.
Related Comments from Research Leaders
A significant incidence of hypersensitivity reactions (HSRs) has been associated with paclitaxel. HSRs first were observed during phase I clinical trials. Prior to the advent of premedications, the incidence was estimated as high as 10%–16% and currently is estimated to be 1%–3%.
Paclitaxel-related HSRs occur rapidly, typically during the first 10 minutes to an hour of the infusion. Signs and symptoms may include any or all of the following: respiratory distress, hypotension, angioedema, f lushing with urticaria, bronchospasm, diaphoresis, hypertension, and chest or back pain. Facial flushing alone is not an indication for stopping the infusion.
— Myers JS.
Clin J Onc Nurs 2000;4(4):161-3.
[Citations omitted]
Docetaxel, despite not being formulated in cremophor, is commonly associated with the development of hypersensitivity reactions. Fortunately, as with paclitaxel, these events rarely result in discontinuation of treatment.
Standard prophylaxis against docetaxel hypersensitivity reactions differs from that used with paclitaxel, patients routinely receiving several oral doses of dexamethasone (for 3-5 days) rather than a single intravenous administration of corticosteroids in combination with histamine-blocking agents. This procedure also protects patients against the development of docetaxel-associated fluid retention. …
For those individuals who develop manifestations of immediate-type hypersensitivity [to paclitaxel] (eg, hypertension, hypotension, diffuse erythema, severe anxiety, dyspnea), it is now recognized that 90% can be successfully treated with the agent if the infusion is quickly discontinued when the initial signs/symptoms are observed (almost always <1-2 min after initiation of the paclitaxel infusion) and then restarted approximately 30 min later.
This is presumably because the initial reaction depletes the immune system of relevant mediators of hypersensitivity (eg, “mast cell degranulation”), which then take some poorly defined period of time to recover sufficiently to result in a subsequent reaction.
— Markman M.
Support Care Cancer 2003;11(3):144-7.
The paclitaxel vehicle CrEL has been shown to influence the toxicity, pharmacokinetics, and antitumor activity of paclitaxel. With regard to paclitaxelinduced HSR [hypersensitivity reactions], CrEL is probably responsible, because other drugs formulated with it produce similar reactions, and CrELfree paclitaxel does not cause HSR.
Likewise, a growing body of evidence shows that CrEL itself is closely related with peripheral neuropathy, one of the main side effects reported for paclitaxel chemotherapy.
— Kim TY et al.
Clin Cancer Res 2004;10(11):3708-16.
EDITOR’S COMMENT
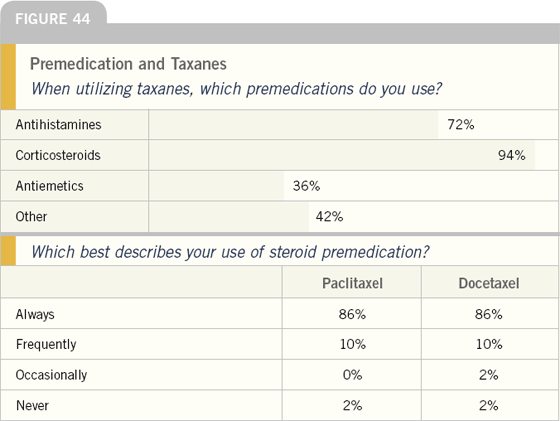
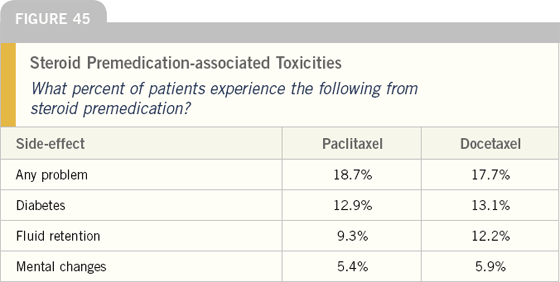
The clinical use of taxanes requires combination preventive agents. The use of corticosteroids is virtually universal. Oncologists estimate that almost 20 percent of patients receiving either docetaxel or paclitaxel experience significant side effects of toxicities associated with the use of premedication with corticosteroids. The most common problem is the onset or exacerbation of diabetes.
Related Comments from Research Leaders
Patients receiving taxane therapy require premedication to minimise the risk of hypersensitivity reactions. However, the premedication guidelines recommended for paclitaxel and docetaxel are markedly different. Patients who receive paclitaxel require both intravenous histamine H1 and H2 antagonists in addition to oral corticosteroids before 1-, 3- or 24-h infusions, although there is some evidence that premedication is not needed before prolonged infusions (those exceeding 96 h).
By contrast, the premedication regimen recommended for patients receiving docetaxel consists of 3 days’ oral dexamethasone (8 mg twice daily).
— Guastalla J et al.
Br J Cancer 2003; 89(Suppl 3):16-22.
Select publications | Previous page
Page 2 of 2
|

